-
Physical Chemistry
Law of Mass action
Law of Mass action – Two Norwegian chemists, Guldberg and Waage, studied experimentally a large number of equilibrium reactions. In…
Read More » -
Organic Chemistry
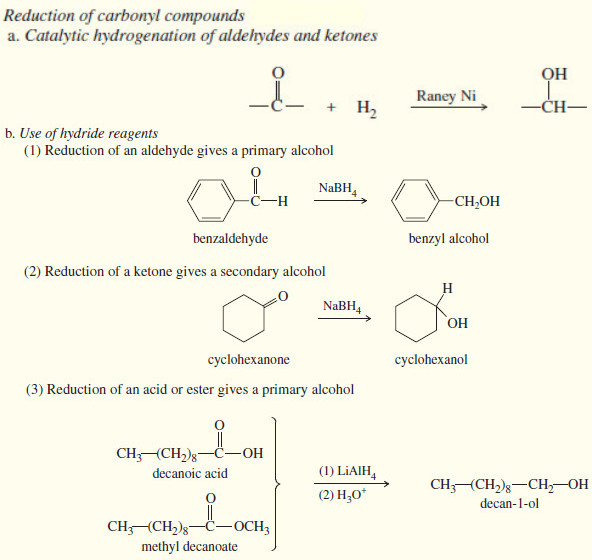
Reduction of the Carbonyl group : Synthesis of Alcohols
Reduction of the Carbonyl group : Synthesis of 1° and 2° Alcohols – Grignard reagents convert carbonyl group to alcohols…
Read More » -
Physical Chemistry
Characteristics of Chemical Equilibrium
Chemical Equilibrium is the state of a reversible reaction when the two opposing reactions occur at the same rate and…
Read More » -
Organic Chemistry
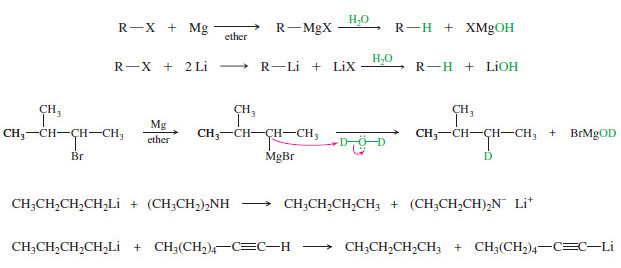
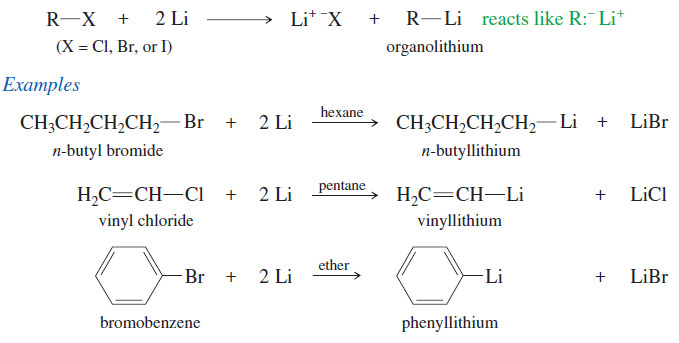
Side Reactions of Organometallic Reagents
Side Reactions of Organometallic Reagents: Reduction of Alkyl Halides – Organometallic Reagents: Grignard and organolithium reagents are strong nucleophiles and…
Read More » -
Organic Chemistry
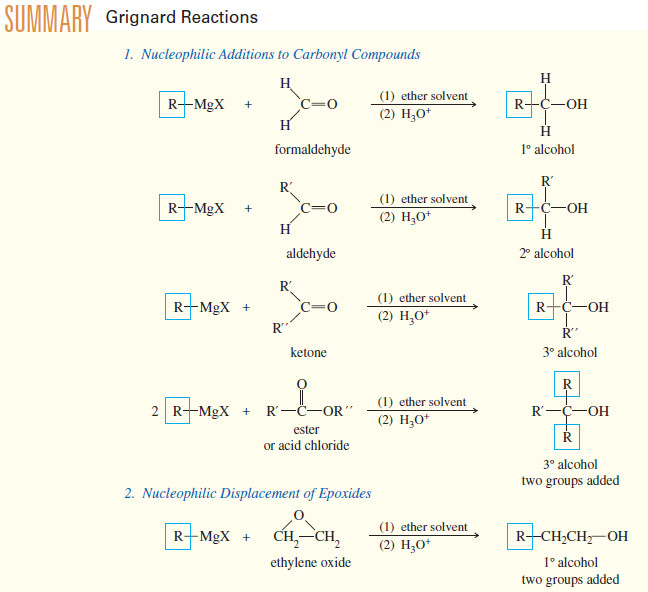
Addition of Grignard Reagents to Carbonyl Compounds
Addition of Organometallic Reagents to Carbonyl Compounds – Because they resemble carbanions, Grignard reagents and organolithium reagents are strong nucleophiles…
Read More » -
Organic Chemistry
Organometallic Reagents for Alcohol Synthesis
Organometallic Reagents for Alcohol Synthesis – Organometallic compounds contain covalent bonds between carbon atoms and metal atoms. – And Organometallic…
Read More » -
Organic Chemistry
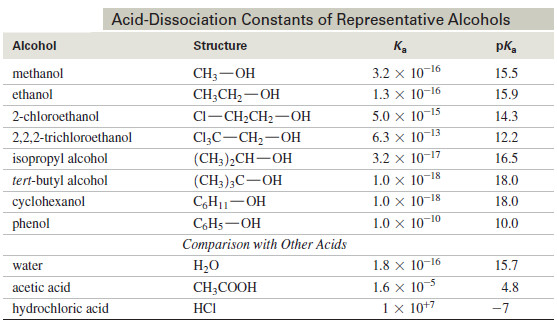
Acidity of Alcohols and Phenols
Acidity of Alcohols and Phenols – we will talk here about some Acidity of Alcohols and Phenols. – Like the…
Read More » -
Organic Chemistry
Commercially Important Alcohols
we will talk about some Commercially Important Alcohols such as: Methanol , Ethanol , isopropyl alcohol (1) Commercially Important Alcohols:…
Read More » -
Organic Chemistry
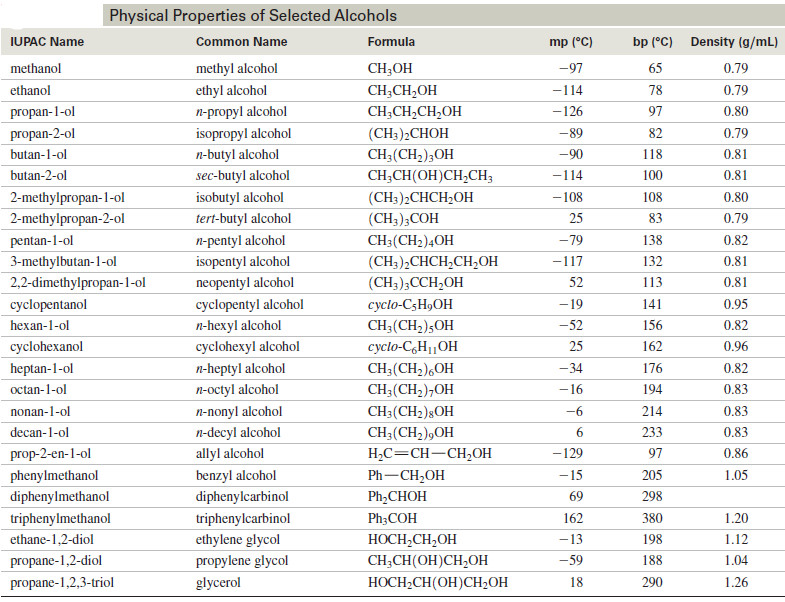
Physical Properties of Alcohols
We will discuss here Physical Properties of Alcohols: (A) Boiling Points of Alcohols (B) Solubility Properties of Alcohols Physical Properties…
Read More » -
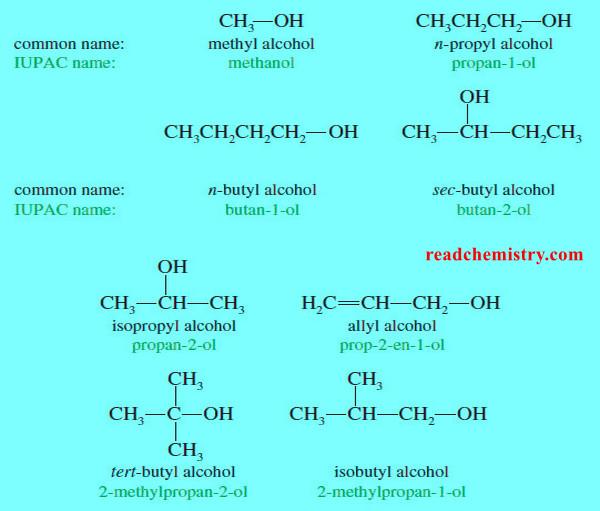
Organic Chemistry
Nomenclature of Alcohols and Phenols
In this subject we will talk about Nomenclature of Alcohols and Phenols (1) Nomenclature of Alcohols: IUPAC Names – The…
Read More » -
Online MCQ
First law of thermodynamics – MCQ online test
Online MCQ test on First law of thermodynamics – In this topic we offer you, online MCQ test in the…
Read More » -
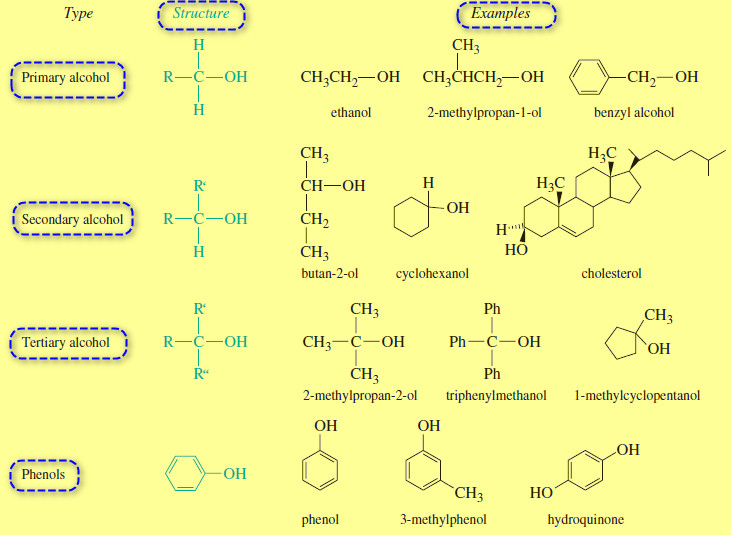
Organic Chemistry
Structure and Classification of Alcohols
– In this subject we will talk about Structure and Classification of Alcohols. What are Alcohols? – Alcohols are organic…
Read More » -
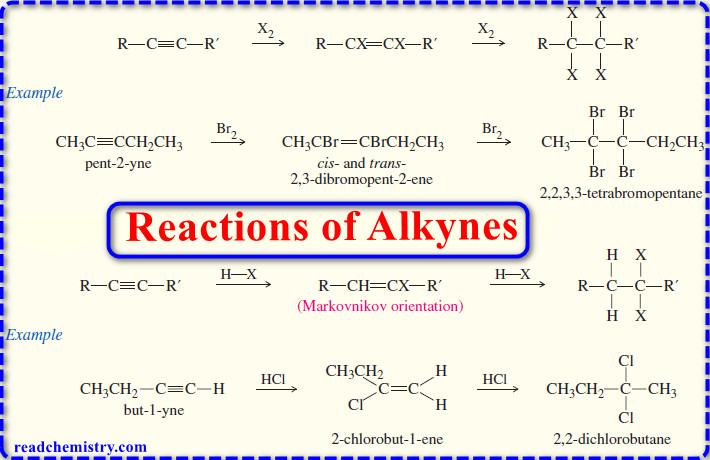
Organic Chemistry
Reactions of Alkynes
Reactions of Alkynes – Many of the reactions of alkynes are similar to the corresponding reactions of alkenes because both…
Read More » -
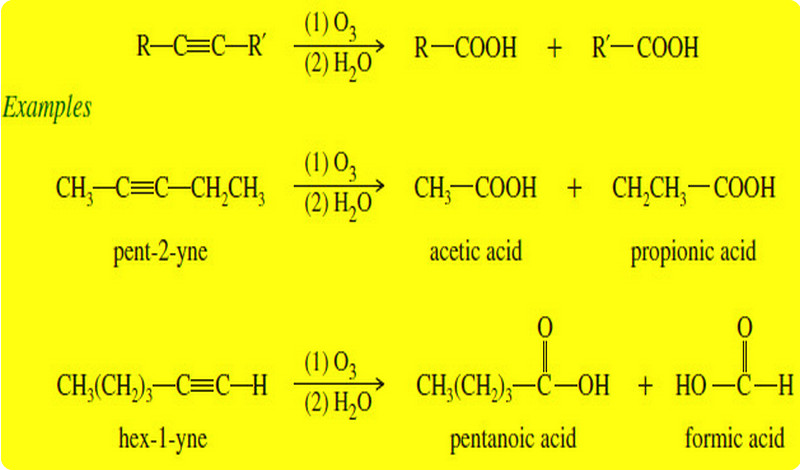
Organic Chemistry
Oxidation of Alkynes
Before we discuss Oxidation of Alkynes we will talk about triple bond of Alkynes What are Alkynes? – Alkynes are…
Read More » -
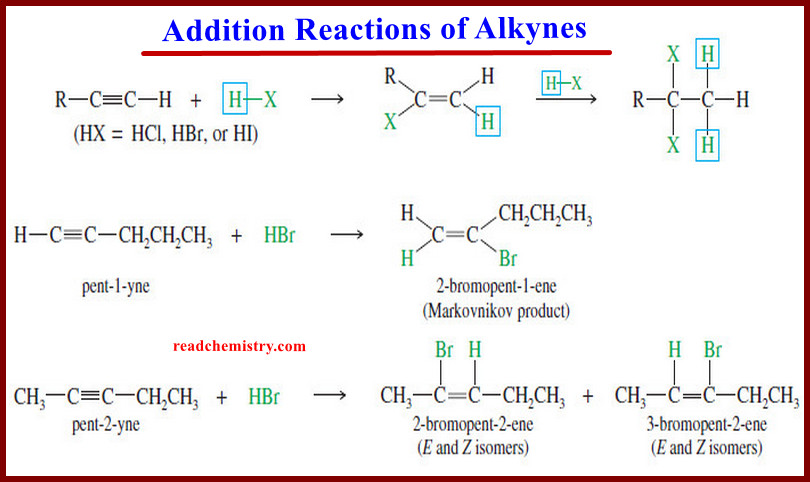
Organic Chemistry
Addition Reactions of Alkynes
Addition Reactions of Alkynes – Many of the reactions of alkynes are similar to the corresponding reactions of alkenes because…
Read More »