General Chemistry
General Chemistry introduces basic chemical concepts, including atomic structure, bonding, reactions, stoichiometry, states of matter, and periodic trends. It lays the groundwork for advanced study in all chemistry branches.
-
MCQ on Chemical Bonding – Orbital Theory
MCQ on Chemical Bonding – Orbital Theory – In this subject, you will find 46 questions and answers MCQ on…
Read More » -
Molecular Orbital Theory
Molecular Orbital Theory – The molecular orbital theory proposed by Hund and Mulliken in 1932 explains the formation of a…
Read More » -
MCQ on Chapter: structure of atom – wave mechanical approach
MCQ on the structure of the atom – In this subject, you will find 50 questions and answers MCQ on…
Read More » -
Electronegativity and Electron Affinity
– In this subject, we will discuss the difference Between Electronegativity and Electron Affinity Electron Affinity – A neutral atom…
Read More » -
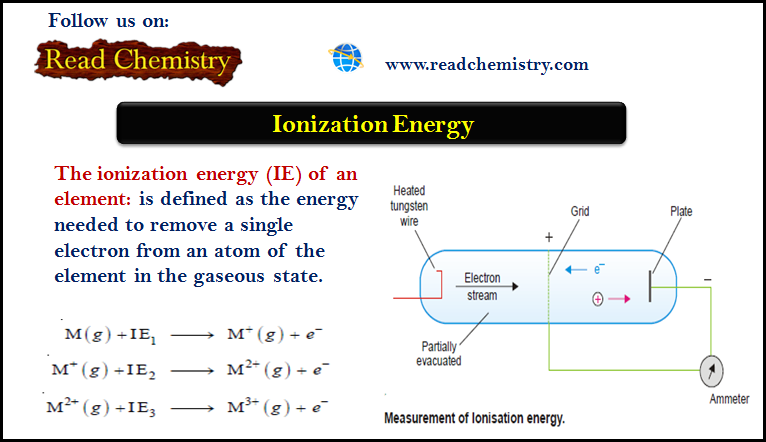
Ionization Energy (Definition – Trends – Measurement)
– The ionization energy (IE) of an element is defined as the energy needed to remove a single electron from…
Read More » -
Electron Configuration Of Elements
– In this subject, we will discuss the rules of Electron Configuration Of Elements Electron Configuration Of Elements – We…
Read More » -
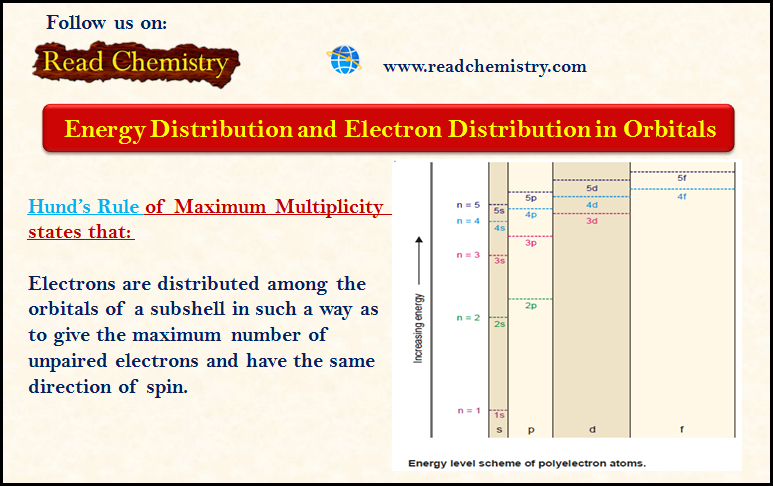
Distribution of Electrons in Orbitals
– In this subject, we will discuss the Distribution of Electrons in Orbitals according to Hund’s Rule. Energy Distribution and…
Read More » -
Pauli Exclusion Principle
– The Pauli exclusion principle is of immense value in telling the maximum number of electrons accommodated in any shell.…
Read More » -
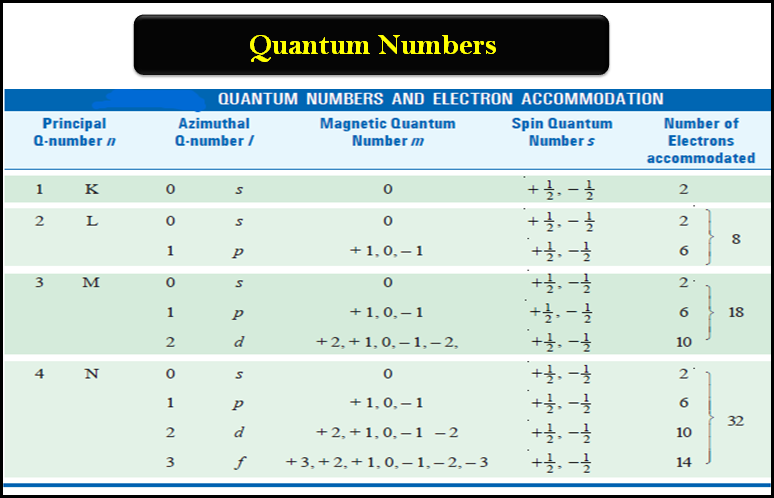
Quantum Numbers (Principal, Azimuthal, Magnetic and Spin)
Quantum Numbers – Bohr’s electronic energy shells or levels, designated as Principal Quantum Numbers (n), could hardly explain the hydrogen…
Read More » -
Schrödinger Wave Equation
Schrödinger Wave Equation – In order to provide sense and meaning to the probability approach, Schrödinger derived an equation known…
Read More » -
Heisenberg’s uncertainty Principle
– Heisenberg’s uncertainty Principle: As it is impossible to know the position and the velocity of any one electron on…
Read More » -
MCQ: Structure of atom – Classical Mechanics
1. In the spectrum of hydrogen atom, the series which falls in ultraviolet region is_______ (a) Lyman series (b) Balmer…
Read More » -
De Broglie Equation – The Wave Nature of Electron
De Broglie Equation – de Broglie had arrived at his hypothesis (de Broglie equation) with the help of Planck’s Quantum…
Read More » -
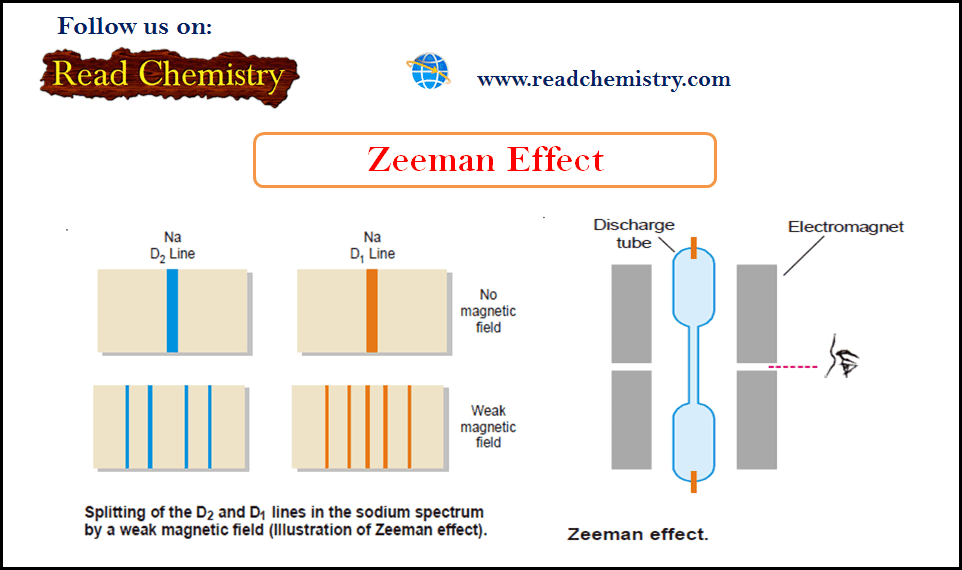
Zeeman Effect
Zeeman Effect – In 1896 Zeeman discovered that spectral lines are split up into components when the source emitting lines…
Read More » -
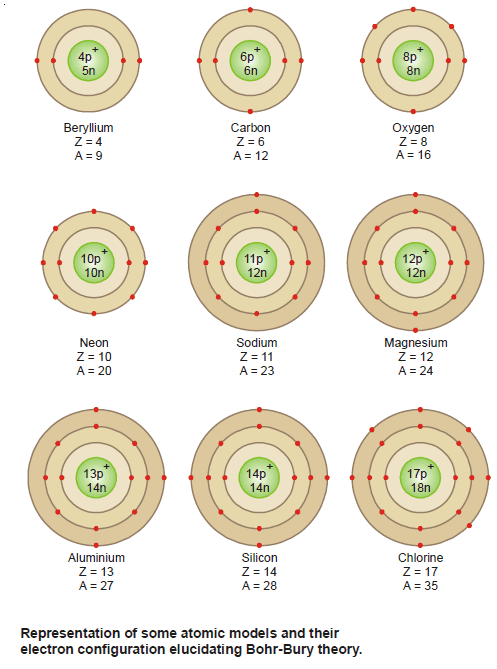
Bohr-Bury Scheme – Electron arrangement in the orbits
– The Bohr-Bury scheme considers that the maximum number of electrons that each orbit can contain is 2 × n2,…
Read More »