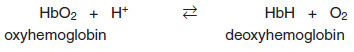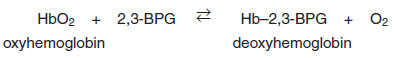Globular Hemeproteins
Globular Hemeproteins
– Hemeproteins are a group of specialized proteins that contain heme as a tightly bound prosthetic group.
– The role of the heme group is dictated by the environment created by the three-dimensional structure of the protein.
– For example, the heme group of a cytochrome functions as an electron carrier that is alternately oxidized and reduced.
– In contrast, the heme group of the enzyme catalase is part of the active site of the enzyme that catalyzes the breakdown of hydrogen peroxide.
– In hemo globin and myoglobin, the two most abundant heme – proteins in humans, the heme group serves to reversibly bind oxygen.
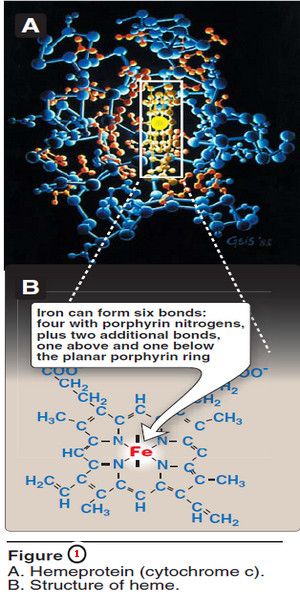
A. Structure of heme
– Heme is a complex of protoporphyrin IX and ferrous iron (Fe2+) (Figure 1).
– The iron is held in the center of the heme molecule by bonds to the four nitrogens of the porphyrin ring.
– The heme Fe2+ can form two additional bonds, one on each side of the planar porphyrin ring.
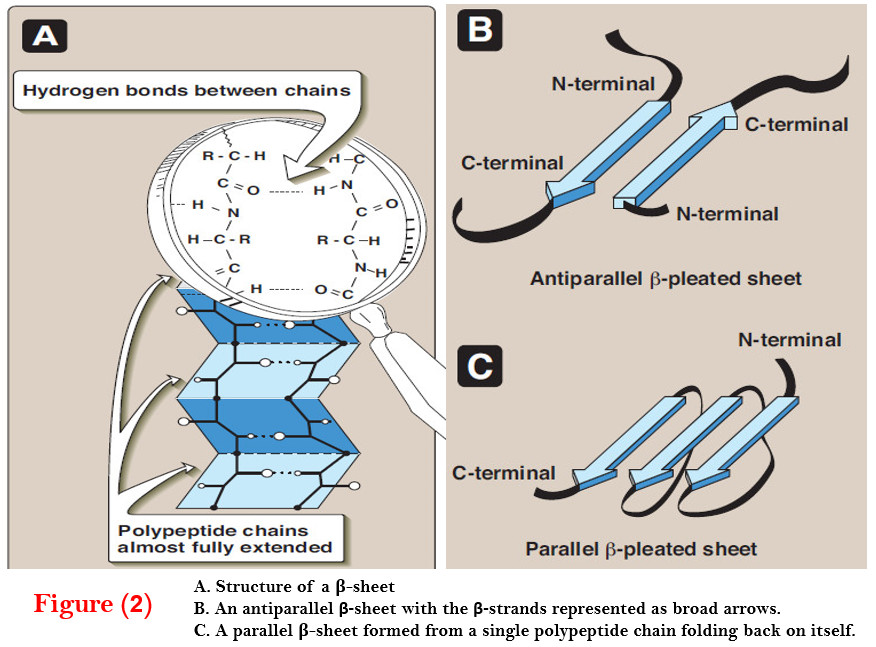
– In myo globin and hemoglobin, one of these positions is coordinated to the side chain of a histidine residue of the globin molecule, whereas the other position is available to bind oxygen (Figure 2).
B. Structure and function of myoglobin
– Myoglobin, a hemeprotein present in heart and skeletal muscle, functions both as a reservoir for oxygen, and as an oxygen carrier that increases the rate of transport of oxygen within the muscle cell.
– Myoglobin consists of a single polypeptide chain that is structurally similar to the individual subunit polypeptide chains of the hemo globin molecule.
– This homology makes myoglobin a useful model for interpreting some of the more complex properties of hemoglobin.
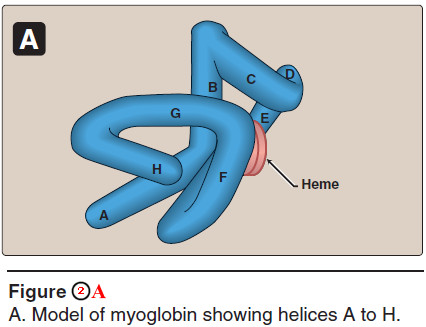
1. α-Helical content
– Myoglobin is a compact molecule, with approximately 80% of its polypeptide chain folded into eight stretches of α-helix.
– These α-helical regions, labeled A to H in Figure 2A, are terminated either by the presence of proline, whose five-membered ring cannot be accommodated in an α helix, or by β-bends and loops stabilized by hydrogen bonds and ionic bonds.
2. Location of polar and nonpolar amino acid residues
– The interior of the myoglobin molecule is composed almost entirely of nonpolar amino acids.
– They are packed closely together, forming a structure stabilized by hydrophobic interactions between these clustered residues.
– In contrast, charged amino acids are located almost exclusively on the surface of the molecule, where they can form hydrogen bonds, both with each other and with water.
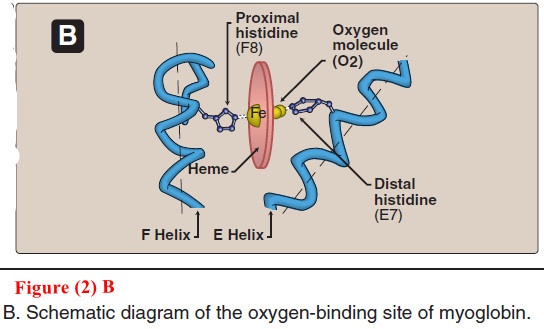
3. Binding of the heme group
– The heme group of myoglobin sits in a crevice in the molecule, which is lined with nonpolar amino acids. Notable exceptions are two histidine residues (Figure 2B).
– One, the proximal histidine (F8), binds directly to the iron of heme.
– The second, or distal histidine (E7), does not directly interact with the heme group, but helps stabilize the binding of oxygen to the ferrous iron.
– The protein, or globin, portion of myoglobin thus creates a special microenvironment for the heme that permits the reversible binding of one oxygen molecule (oxygenation).
– The simultaneous loss of electrons by the ferrous iron (oxidation) occurs only rarely.
C. Structure and function of hemoglobin
– Hemoglobin is found exclusively in red blood cells (RBCs), where its main function is to transport oxygen (O2) from the lungs to the capillariesof the tissues.
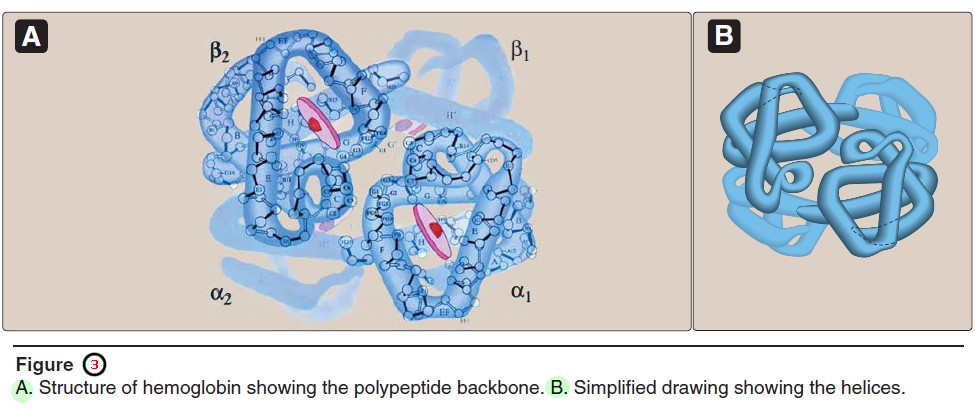
– Hemoglobin A, the major hemoglobin in adults, is composed of four polypeptide chains—two α chains and two β chains—held together by noncovalent interactions (Figure 3).
– Each subunit has stretches of α-helical structure, and a heme-binding pocket similar to that described for myoglobin.
– However, the tetrameric hemoglobin molecule is structurally and functionally more complex than myoglobin.
– For example, hemoglobin can transport H+ and CO2 from the tissues to the lungs, and can carry four molecules of O2 from the lungs to the cells of the body.
– Furthermore, the oxygen-binding properties of hemoglobin are regulated by interaction with allosteric effectors.
1. Quaternary structure of hemoglobin
– The hemoglobin tetramer can be envisioned as being composed of two identical dimers, (αβ)1 and (αβ)2, in which the numbers refer to dimers one and two.
– The two polypeptide chains within each dimer are held tightly together, primarily by hydrophobic interactions (Figure 3.4).
– [Note: In this instance, hydrophobic amino acid residues are localized not only in the interior of the molecule, but also in a region on the surface of each subunit. Interchain hydrophobic interactions form strong associations between α-subunits and β-subunits in the dimers.]
– Ionic and hydrogen bonds also occur between the members of the dimer.
– In contrast, the two dimers are able to move with respect to each other, being held together primarily by polar bonds.
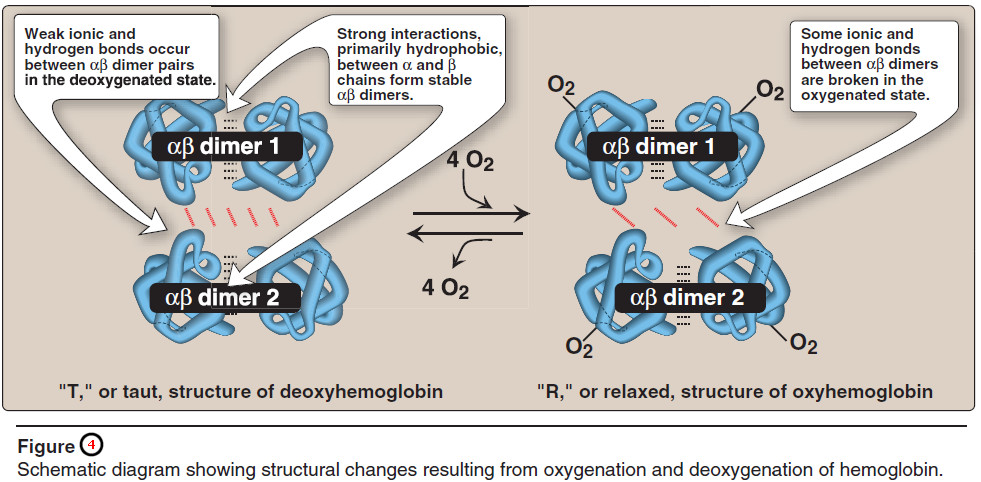
– The weaker interactions between these mobile dimers result in the two dimers occupying different relative positions in deoxyhemoglobin as compared with oxyhemoglobin (see Figure 4).
– [Note: The binding of O2 to the heme iron pulls the iron into the plane of the heme. Because the iron is also linked to the proximal histidine (F8), there is movement of the globin chains that alters the interface between the αβ dimers.]
a. T form
– The deoxy form of hemoglobin is called the “T,” or taut (tense) form. In the T form, the two αβ dimers interact through a network of ionic bonds and hydrogen bonds that constrain the movement of the polypeptide chains.
– The T form is the lowoxygen- affinity form of hemoglobin.
b. R form
– The binding of oxygen to hemoglobin causes the rupture of some of the ionic bonds and hydrogen bonds between the αβ dimers.
– This leads to a structure called the “R,” or relaxed form, in which the polypeptide chains have more freedom of movement (see Figure 4).
– The R form is the highoxygen- affinity form of hemoglobin.
D. Binding of oxygen to myoglobin and hemoglobin
– Myoglobin can bind only one molecule of oxygen, because it contains only one heme group.
– In contrast, hemoglobin can bind four oxygen molecules —one at each of its four heme groups.
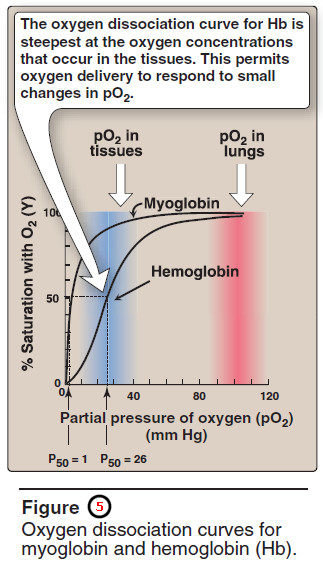
– The degree of saturation (Y) of these oxygen-binding sites on all myoglobin or hemoglobin molecules can vary between zero (all sites are empty) and 100% (all sites are full, Figure 5).
1. Oxygen dissociation curve
– A plot of Y measured at different partial pressures of oxygen (pO2) is called the oxygen dissociation curve.
The curves for myoglobin and hemoglobin show important differences (see Figure 5).
– This graph illustrates that myoglobin has a higher oxygen affinity at all pO2 values than does hemoglobin.
– The partial pressure of oxygen needed to achieve half-saturation of the binding sites (P50) is approximately 1 mm Hg for myoglobin and 26 mm Hg for hemoglobin.
– The higher the oxygen affinity (that is, the more tightly oxygen binds), the lower the P50. [Note: pO2 may also be represented as PO2.]
a. Myoglobin (Mb)
The oxygen dissociation curve for myoglobin has a hyperbolic shape (see Figure 5).
– This reflects the fact that myoglobin reversibly binds a single molecule of oxygen.
– Thus, oxygenated (MbO2) and deoxygenated (Mb) myoglobin exist in a simple equilibrium:
Mb + O2 ↔ MbO2
– The equilibrium is shifted to the right or to the left as oxygen is added to or removed from the system.
– [Note: Myoglobin is designed to bind oxygen released by hemoglobin at the low pO2 found in muscle. Myoglobin, in turn, releases oxygen within the muscle cell in response to oxygen demand.]
b. Hemoglobin (Hb)
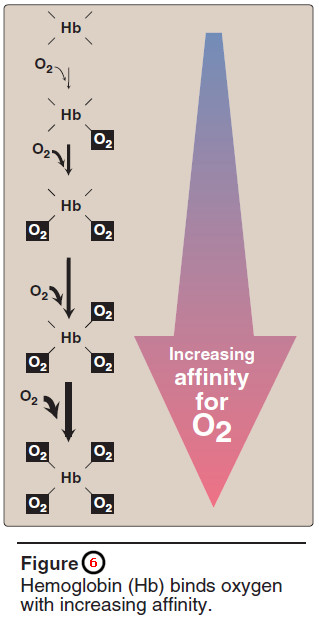
– The oxygen dissociation curve for hemo – globin is sigmoidal in shape (see Figure 5), indicating that the subunits cooperate in binding oxygen.
– Cooperative binding of oxygen by the four subunits of hemoglobin means that the binding of an oxygen molecule at one heme group increases the oxygen affinity of the remaining heme groups in the same hemoglobin molecule (Figure 6).
– This effect is referred to as heme-heme interaction (see below).
– Although it is more difficult for the first oxygen molecule to bind to hemoglobin, the subsequent binding of oxygen occurs with high affinity, as shown by the steep upward curve in the region near 20–30 mm Hg (see Figure 5).
E. Allosteric effects
– The ability of hemoglobin to reversibly bind oxygen is affected by the pO2 (through heme-heme interactions as described above), the pH of the environment, the partial pressure of carbon dioxide, pCO2, and the availability of 2,3-bisphosphoglycerate.
– These are collectively called allosteric (“other site”) effectors, because their interaction at one site on the hemoglobin molecule affects the binding of oxygen to heme groups at other locations on the molecule.
– [Note: The binding of oxygen to myoglobin is not influenced by allosteric effectors.]
1. Heme-heme interactions
– The sigmoidal oxygen dissociation curve reflects specific structural changes that are initiated at one heme group and transmitted to other heme groups in the hemoglobin tetramer.
– The net effect is that the affinity of hemoglobin for the last oxygen bound is approximately 300 times greater than its affinity for the first oxygen bound.
a. Loading and unloading oxygen
– The cooperative binding of oxygen allows hemoglobin to deliver more oxygen to the tissues in response to relatively small changes in the partial pressure of oxygen.
– This can be seen in Figure 5, which indicates pO2 in the alveoli of the lung and the capillaries of the tissues.
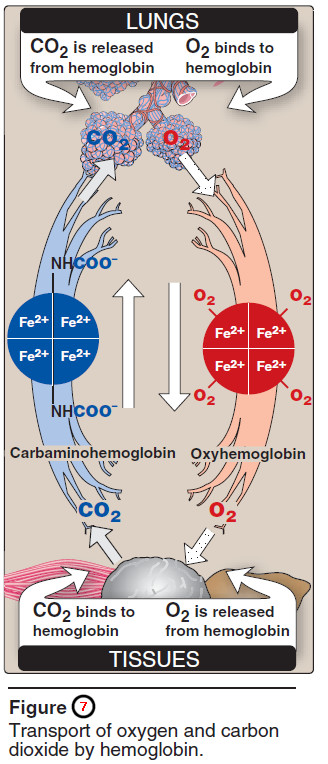
– For example, in the lung, the concentration of oxygen is high and hemoglobin becomes virtually saturated (or “loaded”) with oxygen.
– In contrast, in the peripheral tissues, oxyhemoglobin releases (or “unloads”) much of its oxygen for use in the oxidative metabolism of the tissues (Figure 7).
b. Significance of the sigmoidal oxygen dissociation curve
– The steep slope of the oxygen dissociation curve over the range of oxygen concentrations that occur between the lungs and the tissues permits hemoglobin to carry and deliver oxygen efficiently from sites of high to sites of low pO2.
– A molecule with a hyperbolic oxygen dissociation curve, such as myoglobin, could not achieve the same degree of oxygen release within this range of partial pressures of oxygen.
– Instead, it would have maximum affinity for oxygen throughout this oxygen pressure range and, therefore, would deliver no oxygen to the tissues.
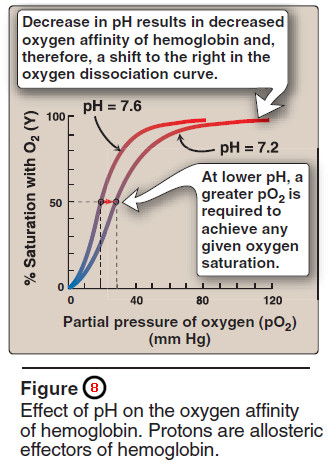
2. Bohr effect
– The release of oxygen from hemoglobin is enhanced when the pH is lowered or when the hemoglobin is in the presence of an increased pCO2.
– Both result in a decreased oxygen affinity of hemoglobin and, therefore, a shift to the right in the oxygen dissociation curve (Figure 8), and both, then, stabilize the T state.
– This change in oxygen binding is called the Bohr effect. Conversely, raising the pH or lowering the concentration of CO2 results in a greater affinity for oxygen, a shift to the left in the oxygen dissociation curve, and stabilization of the R state.
a. Source of the protons that lower the pH
– The concentration of both CO2 and H+ in the capillaries of metabolically active tissues is higher than that observed in alveolar capillaries of the lungs, where CO2 is released into the expired air.
– [Note: Organic acids, such as lactic acid, are produced during anaerobic metabolism in rapidly contracting muscle.]
– In the tissues, CO2 is converted by carbonic anhydrase to carbonic acid:
CO2 + H2O ↔ H2CO3
which spontaneously loses a proton, becoming bicarbonate (the major blood buffer):
H2CO3 ↔ HCO3– + H+
The H+ produced by this pair of reactions contributes to the lowering of pH.
– This differential pH gradient (lungs having a higher pH, tissues a lower pH) favors the unloading of oxygen in the peripheral tissues, and the loading of oxygen in the lung.
– Thus, the oxygen affinity of the hemoglobin molecule responds to small shifts in pH between the lungs and oxygen-consuming tissues, making hemoglobin a more efficient transporter of oxygen
b. Mechanism of the Bohr effect
– The Bohr effect reflects the fact that the deoxy form of hemoglobin has a greater affinity for protons than does oxyhemoglobin.
– This effect is caused by ionizable groups, such as specific histidine side chains that have higher pKas in deoxy hemoglobin than in oxyhemoglobin.
– Therefore, an increase in the concentration of protons (resulting in a decrease in pH) causes these groups to become protonated (charged) and able to form ionic bonds (also called salt bridges).
– These bonds preferentially stabilize the deoxy form of hemoglobin, producing a decrease in oxygen affinity.
– The Bohr effect can be represented schematically as:
where an increase in protons (or a lower pO2) shifts the equilibrium to the right (favoring deoxyhemoglobin), whereas an increase in pO2 (or a decrease in protons) shifts the equilibrium to the left.
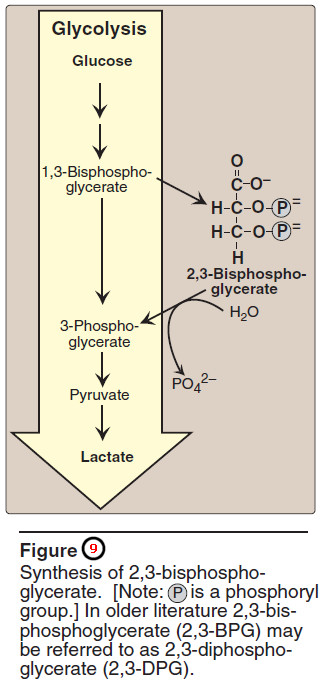
3. Effect of 2,3-bisphosphoglycerate on oxygen affinity
– 2,3-Bis – phospho glycerate (2,3-BPG) is an important regulator of the binding of oxygen to hemoglobin.
– It is the most abundant organic phosphate in the RBC, where its concentration is approximately that of hemoglobin.
– 2,3-BPG is synthesized from an intermediate of the glycolytic pathway (Figure 9)
a. Binding of 2,3-BPG to deoxyhemoglobin
– 2,3-BPG decreases the oxygen affinity of hemoglobin by binding to deoxy – hemoglobin but not to oxyhemoglobin.
– This preferential binding stabilizes the taut conformation of deoxyhemoglobin.
– The effect of binding 2,3-BPG can be represented schematically as:
b. Binding site of 2,3-BPG
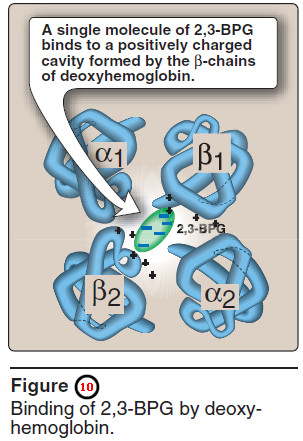
– One molecule of 2,3-BPG binds to a pocket, formed by the two β-globin chains, in the center of the deoxyhemoglobin tetramer (Figure 10).
– This pocket contains several positively charged amino acids that form ionic bonds with the negatively charged phosphate groups of 2,3-BPG.
– [Note: A mutation of one of these residues can result in hemoglobin variants with abnormally high oxygen affinity.]
– 2,3-BPG is expelled on oxygenation of the hemoglobin.
c. Shift of the oxygen dissociation curve
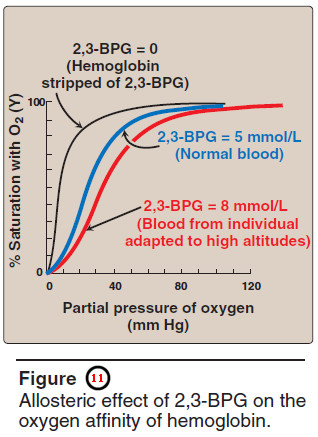
– Hemoglobin from which 2,3-BPG has been removed has a high affinity for oxygen.
– However, as seen in the RBC, the presence of 2,3-BPG significantly reduces the affinity of hemoglobin for oxygen, shifting the oxygen dissociation curve to the right (Figure 11).
– This reduced affinity enables hemoglobin to release oxygen efficiently at the partial pressures found in the tissues.
d. Response of 2,3-BPG levels to chronic hypoxia or anemia
– The concentration of 2,3-BPG in the RBC increases in response to chronic hypoxia, such as that observed in chronic obstructive pulmonary disease (COPD) like emphysema, or at high altitudes, where circulating hemoglobin may have difficulty receiving sufficient oxygen.
– Intracellular levels of 2,3-BPG are also elevated in chronic anemia, in which fewer than normal RBCs are available supply the body’s oxygen needs.
– Elevated 2,3-BPG levels lower the oxygen affinity of hemo – globin, permitting greater unloading of oxygen in the capillaries of the tissues (see Figure 11).
e. Role of 2,3-BPG in transfused blood
– 2,3-BPG is essential for the normal oxygen transport function of hemoglobin.
– However, storing blood in the currently available media results in a decrease in 2,3-PBG.
– Stored blood displays an abnormally high oxygen affinity, and fails to unload its bound oxygen properly in the tissues.
– Hemoglobin deficient in 2,3-BPG thus acts as an oxygen “trap” rather than as an oxygen transport system.
– Transfused RBCs are able to restore their depleted supplies of 2,3-BPG in 6–24 hours. However, severely ill patients may be compromised if transfused with large quantities of such 2,3 BPG–“stripped” blood.
– [Note: The maximum storage time for red cells has been doubled (21 to 42 days, with median time of 15 days) by changes in H+, phosphate and hexose sugar concentration, and by the addition of adenine . Although the content of 2,3-BPG was not greatly affected by these changes, ATP production was increased and improved RBC survival.]
4. Binding of CO2
Most of the CO2 produced in metabolism is hydrated and transported as bicarbonate ion. However, some CO2 is carried as carbamate bound to the N-terminal amino groups of hemoglobin (forming carbaminohemoglobin, see Figure 3.7), which can be represented schematically as follows:
Hb – NH2 + CO2 ↔ Hb – NH – COO– + H+
– The binding of CO2 stabilizes the T (taut) or deoxy form of hemoglobin, resulting in a decrease in its affinity for oxygen and a right shift in the oxygen dissociation.
– In the lungs, CO2 dissociates from the hemoglobin, and is released in the breath.
5. Binding of CO
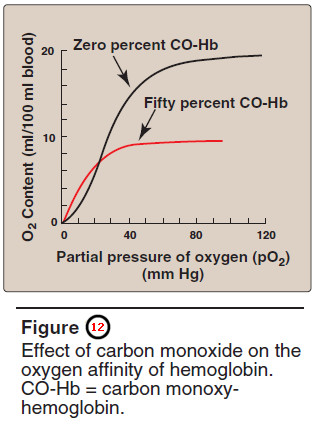
– Carbon monoxide (CO) binds tightly (but reversibly) to the hemoglobin iron, forming carbon monoxy hemo – globin (or carboxyhemoglobin).
– When CO binds to one or more of the four heme sites, hemoglobin shifts to the relaxed conformation, causing the remaining heme sites to bind oxygen with high affinity.
– This shifts the oxygen dissociation curve to the left, and changes the normal sigmoidal shape toward a hyperbola.
– As a result, the affected hemoglobin is unable to release oxygen to the tissues (Figure 12).
– [Note: The affinity of hemoglobin for CO is 220 times greater than for oxygen.
– Consequently, even minute concentrations of CO in the environment can produce toxic concentrations of carbon monoxyhemoglobin in the blood.
– For example ple, increased levels of CO are found in the blood of tobacco smokers.
– Carbon monoxide toxicity appears to result from a combination of tissue hypoxia and direct CO-mediated damage at the cellular level.]
– Carbon monoxide poisoning is treated with 100% oxygen at high pressure (hyperbaric oxygen therapy), which facilitates the dissociation of CO from the hemoglobin.
– [Note: CO inhibits Complex IV of the electron transport chain.]
– In addition to O2, CO2, and CO, nitric oxide gas (NO) also is carried by hemoglobin.
– NO is a potent vasodilator. It can be taken up (salvaged) or released from RBCs, thus modulating NO availability and influencing vessel diameter.
F. Minor hemoglobins
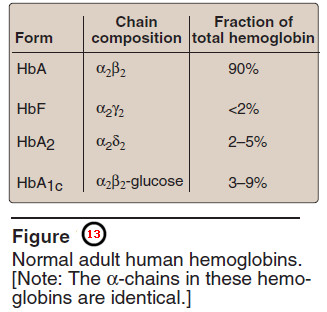
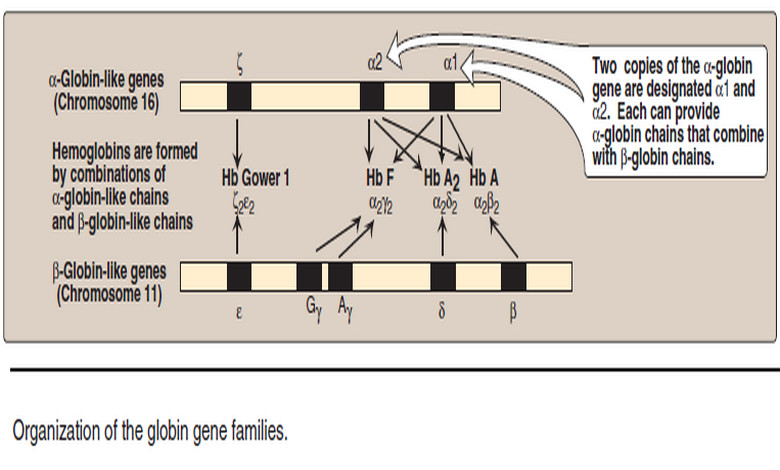
– It is important to remember that human hemoglobin A (Hb A) is just one member of a functionally and structurally related family of proteins, the hemoglobins (Figure 13).
– Each of these oxygen-carrying proteins is a tetramer, composed of two α-globin polypeptides and two β-globin (or β-globin-like) polypeptides.
– Certain hemoglobins, such as Hb F, are normally synthesized only during fetal development, whereas others, such as Hb A2, are synthesized in the adult, although at low levels compared with Hb A.
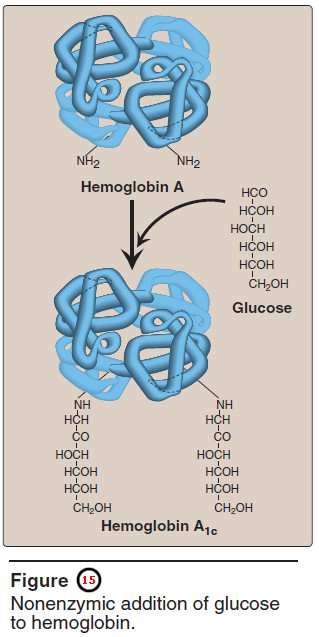
– Hb A can also become modified by the covalent addition of a hexose.
– For example, addition of glucose forms the glucosylated hemoglobin derivative, Hb A1c.
1. Fetal hemoglobin (Hb F)
– Hb F is a tetramer consisting of two α chains identical to those found in Hb A, plus two γ chains (α2γ2, see Figure 13).
– The γ chains are members of the β-globin gene family.
a. Hb F synthesis during development
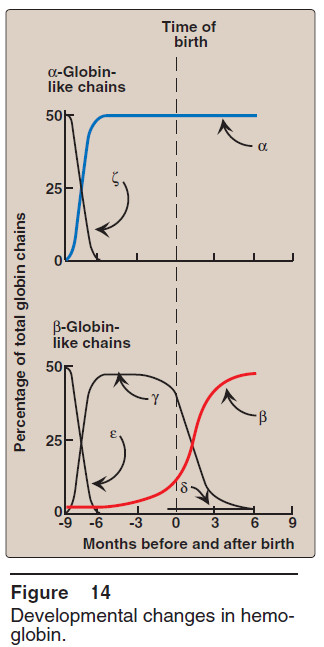
– In the first month after conception, embryonic hemoglobins such as Hb Gower 1, composed of two α-like zeta (ζ) chains and two β-like epsilon (ε) chains (ζ2ε2), are synthesized by the embryonic yolk sac.
– In the fifth week of gestation, the site of globin synthesis shifts, first to the liver and then to the marrow, and the primary product is Hb F.
– Hb F is the major hemoglobin found in the fetus and newborn, accounting for about 60% of the total hemoglobin in the erythrocytes during the last months of fetal life (Figure 14).
– Hb A synthesis starts in the bone marrow at about the eighth month of pregnancy and gradually replaces Hb F.
– (Figure 14 shows the relative production of each type of hemoglobin chain during fetal and postnatal life.)
– [Note: Hb F represents less than 1% of the Hb in most adults, and is concentrated in RBCs known as F cells.]
b. Binding of 2,3-BPG to Hb F
– Under physiologic conditions, Hb F has a higher affinity for oxygen than does Hb A, as a result of Hb F binding only weakly to 2,3-BPG.
– [Note: The γ-globin chains of Hb F lack some of the positively charged amino acids that are responsible for binding 2,3-BPG in the β-globin chains.]
– Because 2,3-BPG serves to reduce the affinity of hemoglobin for oxygen, the weaker interaction between 2,3-BPG and Hb F results in a higher oxygen affinity for Hb F relative to Hb A.
– In contrast, if both Hb A and Hb F are stripped of their 2,3-BPG, they then have a similar affinity for oxygen
– The higher oxygen affinity of Hb F facilitates the transfer of oxygen from the maternal circulation across the placenta to the RBCs of the fetus.
2. Hemoglobin A2 (Hb A2)
– Hb A2 is a minor component of normal adult hemoglobin, first appearing shortly before birth and, ultimately, constituting about 2% of the total hemoglobin.
– It is composed of two α-globin chains and two δ-globin chains (α2δ2, see Figure 13).
3. Hemoglobin A1c (HbA1c)
– Under physiologic conditions, Hb A is slowly and nonenzymically glycosylated, the extent of glycosylation being dependent on the plasma concentration of a particular hexose.
– The most abundant form of glycosylated hemoglobin is HbA1c.
– It has glucose residues attached predominantly to the NH2 groups of the N-terminal valines of the β-globin chains (Figure 15).
– Increased amounts of Hb A1c are found in RBCs of patients with diabetes mellitus, because their Hb A has contact with higher glucose concentrations during the 120-day lifetime of these cells.