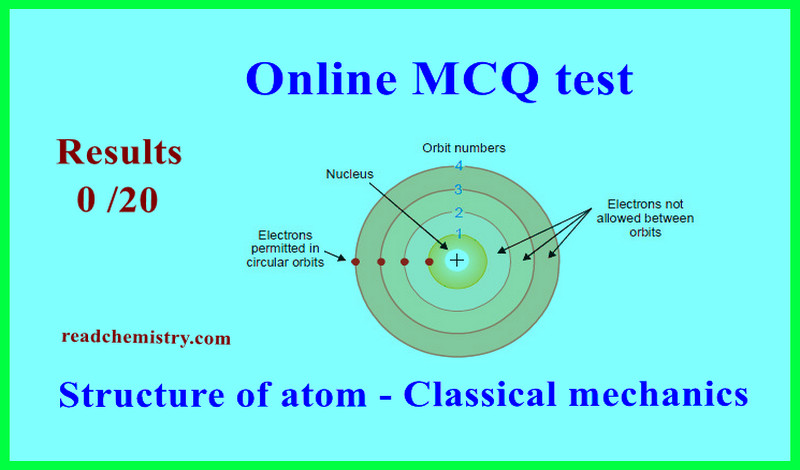
Gaseous state – Online MCQ test
Online MCQ test on gaseous state
– In this topic we offer you, online MCQ test in the fundmental of gaseous state .
– It is a simple test to measure the strength of your knowledge in the basics of understanding the topics of gaseous state.
– The test consists of only (20) questions (multiple choice) you must answer all of them so that your final score appears automatically at the top of the topic.
– you will pass the Exam If get 70% or more.
– If you fail the test, you can retake the test again by reloading the page
– Let’s start the test, good luck.
Starting the test of gaseous state
Results
Congratulation
Try again
#1. Which of the following is the correct mathematical relation for Charle’s law at constant pressure?
#2. According to Boyle’s law the volume of a fixed mass of a gas, at constant temperature, is
#3. 290 ml of a gas at 17°C is cooled to –13°C at constant pressure. The new volume of the gas will be
#4. Which of the following gases will have the highest rate of diffusion?
#5. For one mole of a gas, the ideal gas equation is ____
#6. The compressibility factor, z, for an ideal gas is ____
#7. In van der Waal’s equation of state for a non-ideal gas the net force of attraction among the molecules is given by ____
#8. Equal volume of all gases at the same temperature and pressure contain equal number of molecules” is the statement of _____
#9. The total pressure of a mixture of gases (non–reacting) is equal to the sum of the partial pressures of all the gases present” is the statement of ____
#10. The free path is the distance travelled by the molecule ____
#11. According to Gay Lussac’s law for a fixed volume of a given gas?
#12. Which of the following gases will have the lowest rate of diffusion?
#13. For one mole of a gas the kinetic energy is given by ___
#14. The average velocity of a gas is given by _____
#15. The value of γ , the specific heat ratio, for a monoatomic gas is _____
#16. The real gases show nearly ideal behaviour at _____
#17. Gas A diffuses twice as fast as another gas B. If the vapour density of the gas A is 2, the molecular mass of gas B is __
#18. The mass of 2240 ml of CO2 at NTP will be _____
#19. A gas is heated at constant temperature. Then ____
#20. If the pressure and absolute temperature of 3 litres of a gas are doubled, its volume would be ___
Click on the word (Finish) to see the test result
MCQs test on Chemistry
– Our multiple choice questions and answers (MCQs) focus on all areas of Chemistry covering all chemistry topics.
– One can read MCQs on Chemistry here. These topics are chosen from a collection of the most authoritative and best reference books on Chemistry.
– One should spend 1 hour daily practicing these MCQs for 2-3 months to learn and assimilate Chemistry subject comprehensively.
– This way of systematic learning will prepare anyone easily for Chemistry – Class exams, contests, online tests, quizzes, MCQ-tests, viva-voce, interviews, and certifications.
– These MCQs are organized chapterwise and each Chapter is futher organized topicwise.
– Every MCQ set focuses on a specific topic of Chapter in Chemistry.
Who should Practice MCQ Chemistry ?
(1) Students who are preparing for various school tests such as unit-tests, term-tests, semester tests, half-yearly and yearly tests and examinations on Chemistry
(2) Students who are preparing for Online/Offline Tests/Contests in Chemistry.
(3) Students who wish to sharpen their knowledge of Chemistry.
(4) Students who are studying in CBSE, ICSE, IGCSE schools as well as various State Board schools.
(5) Students who are preparing for competitive examinations such as NTSE, Olympiad.
(6) Students who are preparing for Central board examinations in Chemistry namely CBSE, ICSE, and NIOS Board.
(7)Students who are preparing for State board examinations in Chemistry such as PUC, Intermediate exams, etc.
(8) Students who are preparing for engineering and medical entrance examinations like NEET, IIT JEE, AIIMS, JIPMER & SAT Exams in Chemistry.
Get more: MCQ online test in Chemistry by English
Get more: MCQ online test in Chemistry by Arabic