-
Organic Chemistry
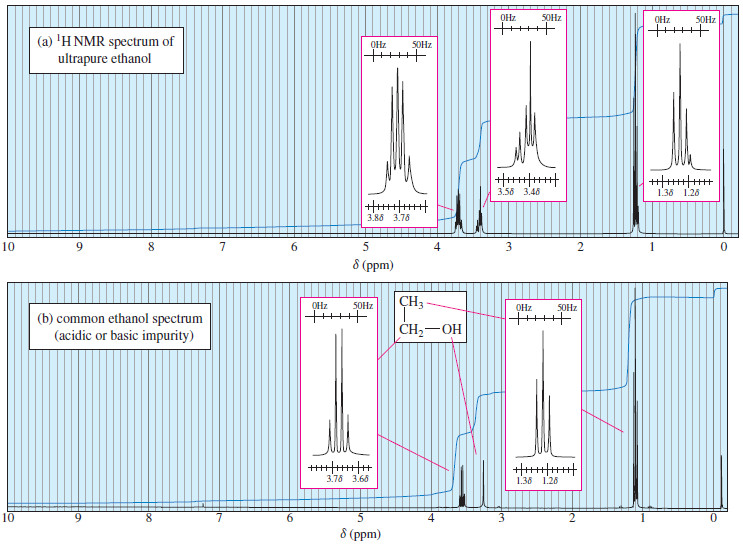
Time Dependence of NMR Spectroscopy
– In this topic, we will discuss The Time Dependence of NMR Spectroscopy. Time Dependence of NMR Spectroscopy – We…
Read More » -
Analytical Chemistry
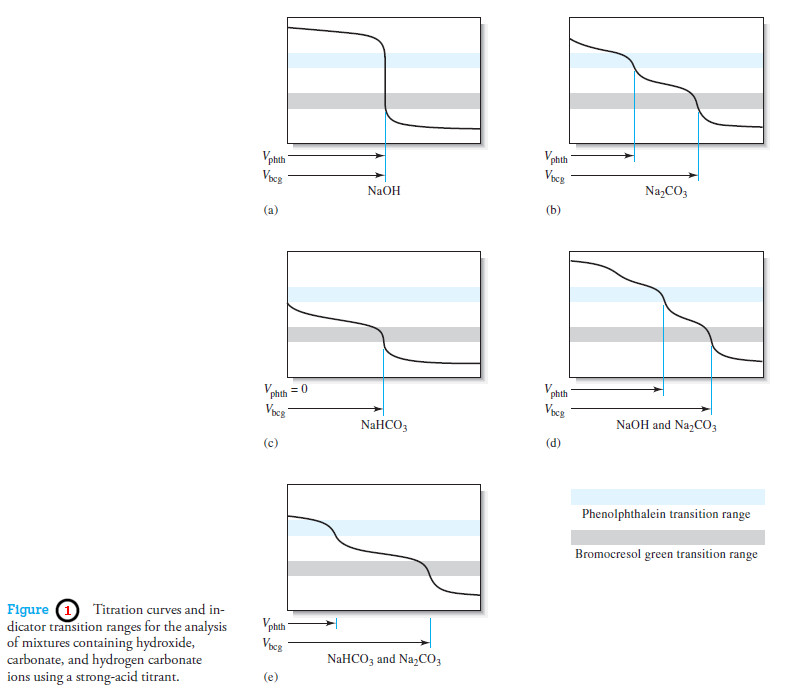
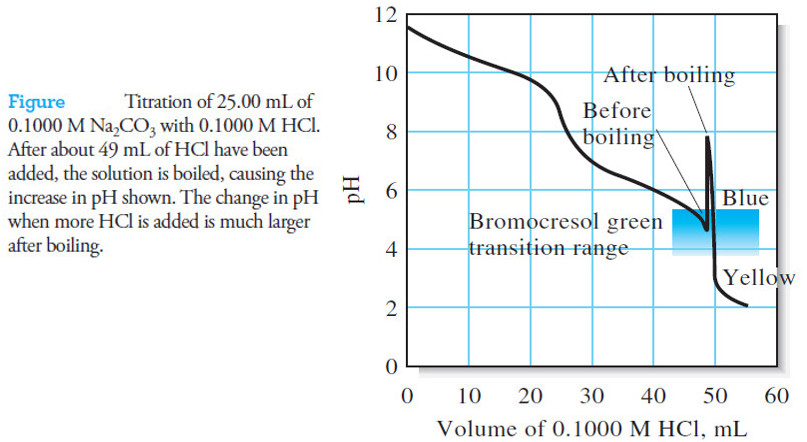
Applications of Neutralization Titrations
– In this topic, we will discuss The Applications of Neutralization Titrations. Typical Applications of Neutralization Titrations – Neutralization titrations…
Read More » -
Organic Chemistry
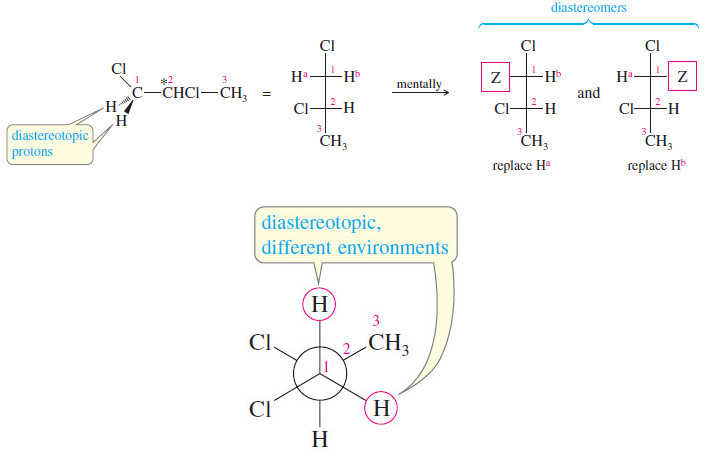
Stereochemical Nonequivalence of Protons in NMR Spectroscopy
– In the this topic we talk about Stereochemical Nonequivalence of Protons in NMR Spectroscopy. Stereochemical Nonequivalence of Protons –…
Read More » -
Physical Chemistry
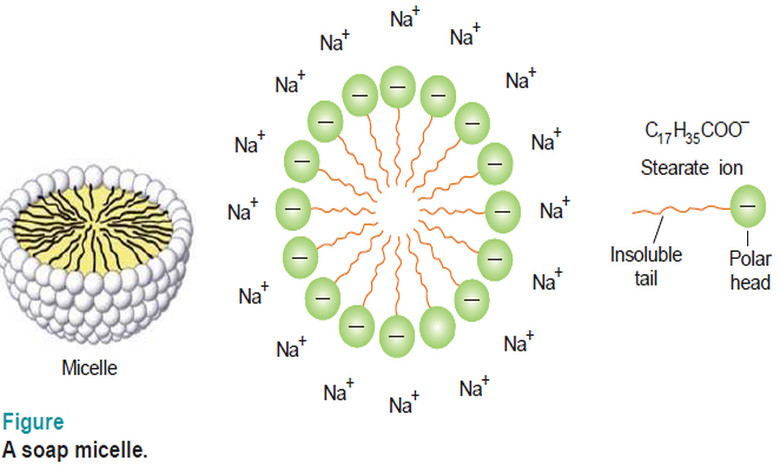
Associated Colloids
– In this subject, we will discuss the Associated Colloids. Associated Colloids – The molecules of substances as soaps and…
Read More » -
Physical Chemistry
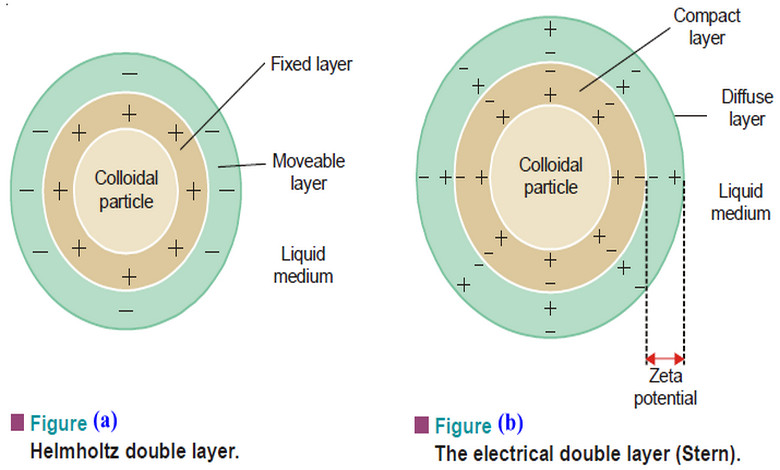
Electrical Properties of Sols
Electrical Properties of Sols – In this topic we will discuss the Electrical Properties of Sols as follow: (1) The…
Read More » -
Analytical Chemistry
Reagents for Neutralization Titrations
– In this subject, we will discuss the Reagents for Neutralization Titrations. Reagents for Neutralization Titrations – we noted before…
Read More » -
Physical Chemistry
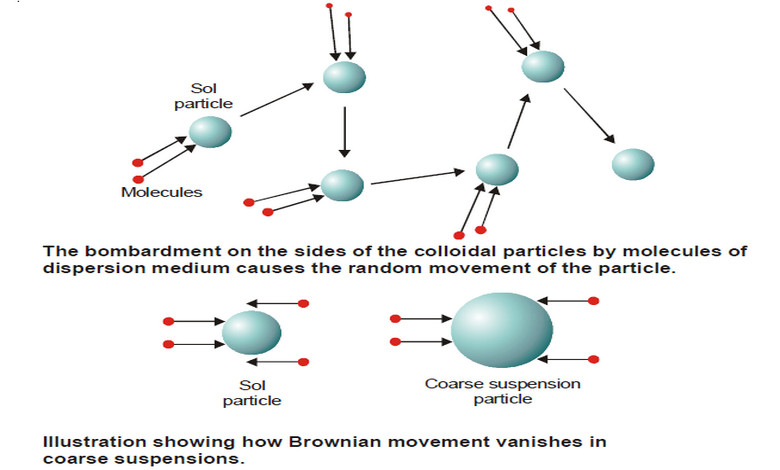
Kinetic Properties of Sols – Brownian movement
Kinetic Properties of Sols – In this topic, we will discuss Kinetic Properties of Sols. Brownian Movement – When a…
Read More » -
Physical Chemistry
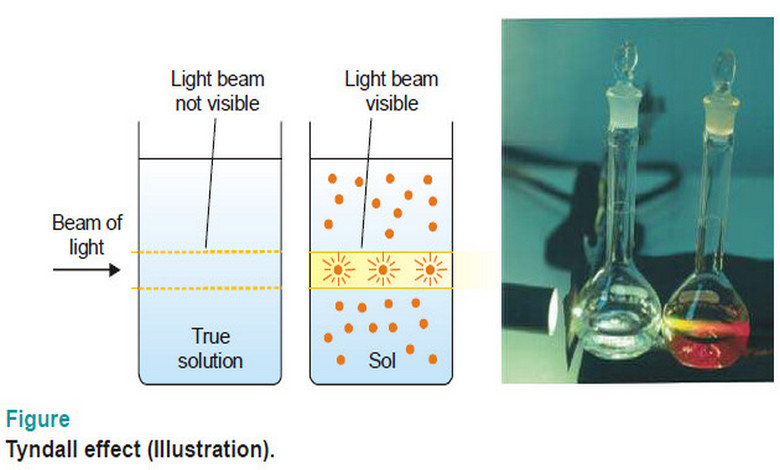
Optical Properties of Sols
Optical Properties of Sols – In this topic, we will discuss Optical Properties of Sols as follow: Sols exhibit Tyndall…
Read More » -
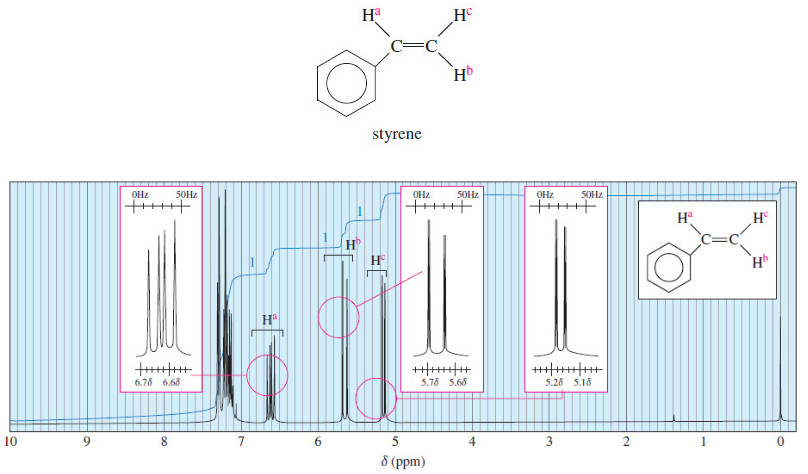
Organic Chemistry
Complex Splitting in ¹H NMR Spectra
– In the last topic we talk about Spin-Spin Splitting in ¹H NMR Spectra, but In this topic, we will…
Read More » -
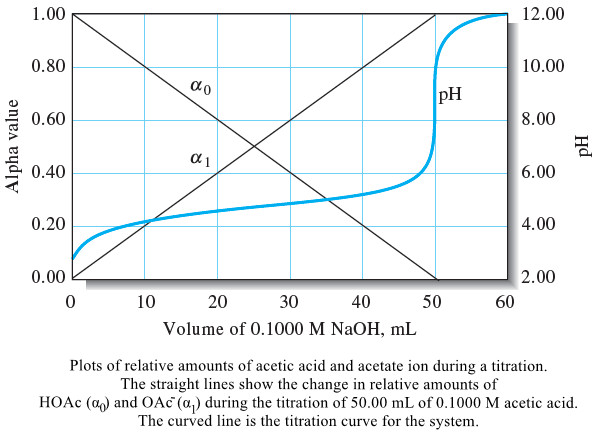
Analytical Chemistry
The Composition of Solutions During acid/Base Titration
– In this topic, we will discuss The Composition of Solutions During acid/Base Titration. The Composition of Solutions During acid/Base…
Read More » -
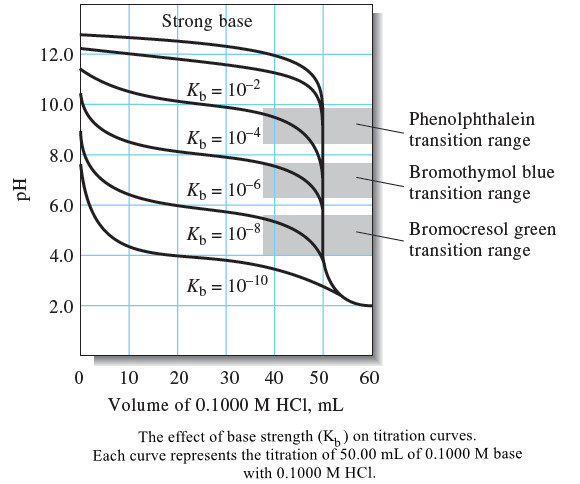
Analytical Chemistry
Titration Curves for Weak Bases
– In this topic, we will discuss The titration Curves for Weak Bases. Titration Curves for Weak Bases – The…
Read More » -
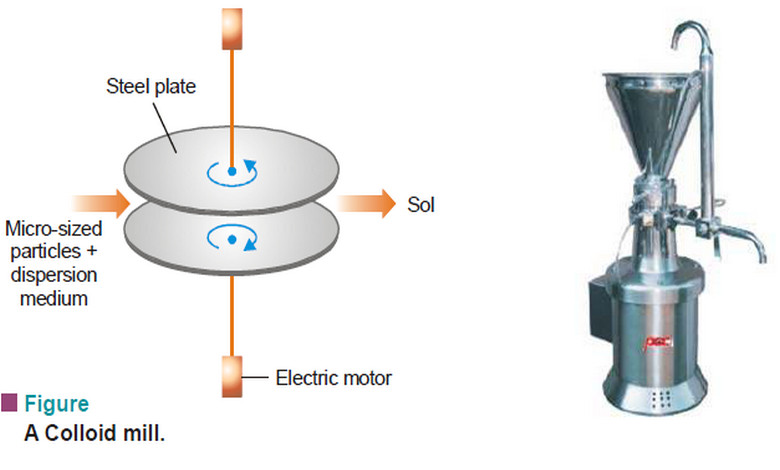
Physical Chemistry
Preparation of Sols and Purification of Sols
– In this topic, we will discuss Preparation of Sols, Purification of Sols and its colors Preparation of Sols –…
Read More » -
Analytical Chemistry
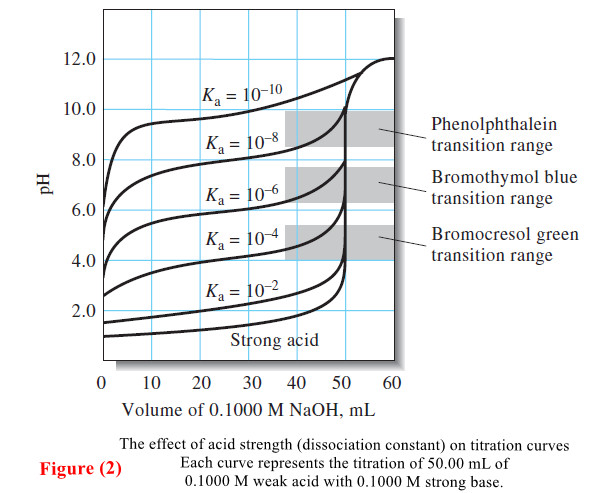
Titration Curves for Weak Acids
Titration Curves for Weak Acids – Four distinctly different types of calculations are needed to compute values for a weak…
Read More » -
Organic Chemistry
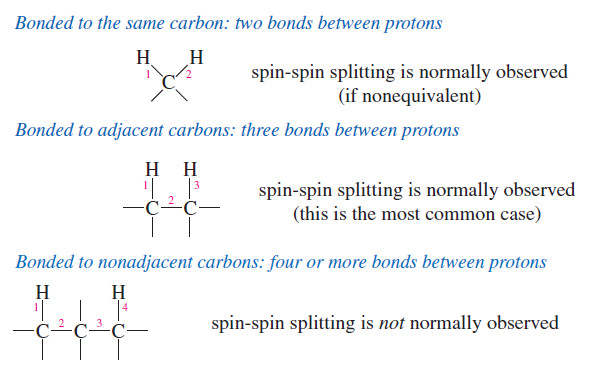
Spin-Spin Splitting in ¹H NMR Spectra
– In this topic, we will discuss The Spin-Spin Splitting in ¹H NMR Spectra. Theory of Spin-Spin Splitting – A…
Read More » -
Physical Chemistry
Lyophilic and Lyophobic sols : Defination, Properties, Comparison
– In this topic, we will discuss The Lyophilic and Lyophobic sols : Defination, Characteristics, and Comparison. Defination of Lyophilic…
Read More »