Physical Chemistry blends chemistry with physics, studying matter’s behavior using concepts like thermodynamics, kinetics, quantum mechanics, and spectroscopy. It explains how and why chemical reactions occur at the molecular level.
Physical Chemistry
-
Hess’s Law ( statement, Illustration, application, Problems)
– Hess’s Law may be stated as: (If a chemical change can be made to take place in two or…
Read More » -
Energy Changes During Transitions or Phase Changes
– In this subject, we will discuss Energy Changes During Transitions or Phase Changes. Energy Changes During Transitions or Phase…
Read More » -
Heat of Combustion (Definition, Applications, Solved Problems)
Heat of Combustion – The heat of combustion of a substance is defined as The change in enthalpy of…
Read More » -
Heat of Formation (Definition, Applications, Solved Problems)
Heat of Formation – The heat of formation of a compound is defined as The change in enthalpy that takes…
Read More » -
Different Types of Heat of Reaction (Enthalpy)
– The heat of reaction or enthalpy changes accompanying chemical reactions are expressed in different ways, depending on the nature…
Read More » -
Variation of heat of reaction with temperature
– In this subject, the Variation of heat of reaction with temperature will be discussed. Heat of Reaction or Enthalpy…
Read More » -
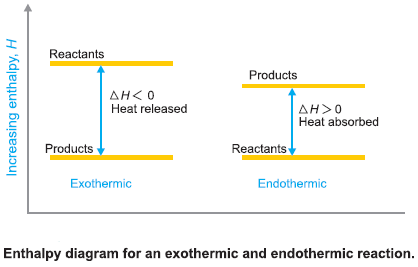
Enthalpy of Reaction
– For reactions involving solids and liquids only the change in volume (ΔV) is very small and the term P…
Read More » -
MCQ on the First law of Thermodynamics
MCQ on the First Law of Thermodynamics – In this subject, you will find 50 questions and answers MCQ on…
Read More » -
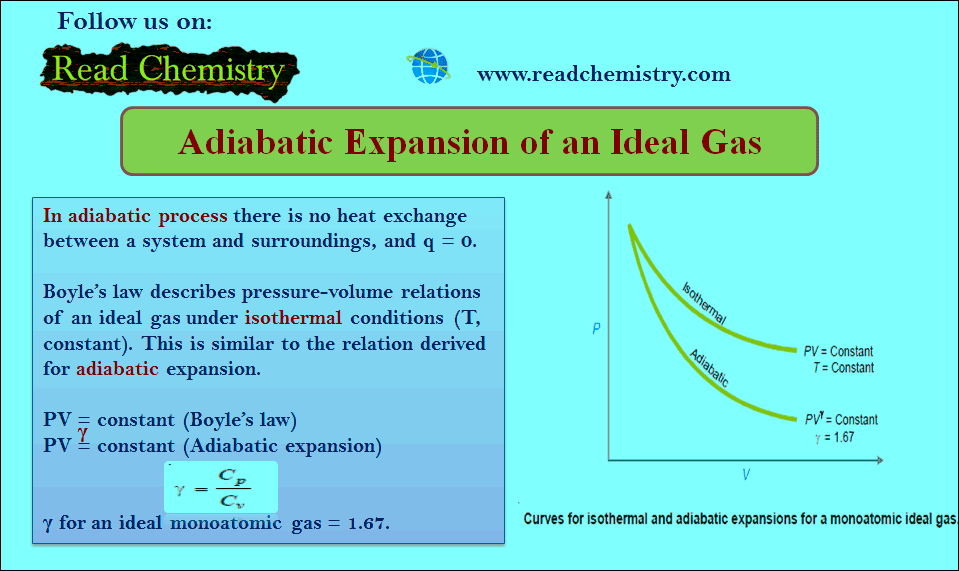
Adiabatic Expansion of an Ideal Gas
Adiabatic Expansion of an Ideal Gas – A process carried in a vessel whose walls are perfectly insulated so that…
Read More » -
Heat Capacity – Molar Heat Capacity
Molar Heat Capacity – By heat capacity of a system, we mean the capacity to absorb heat and store energy.…
Read More » -
Enthalpy of A System
– Enthalpy (H) is the total heat content of a system at constant pressure and is equivalent to the internal…
Read More » -
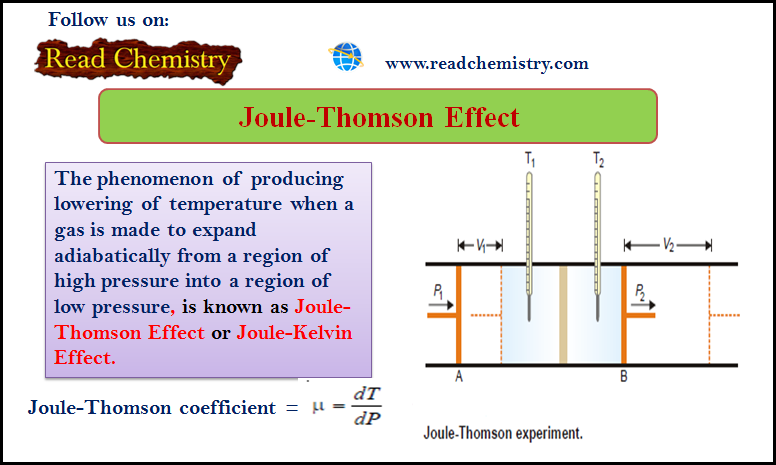
Joule-Thomson Effect
– The phenomenon of producing a lowering of temperature when a gas is made to expand adiabatically from a region…
Read More » -
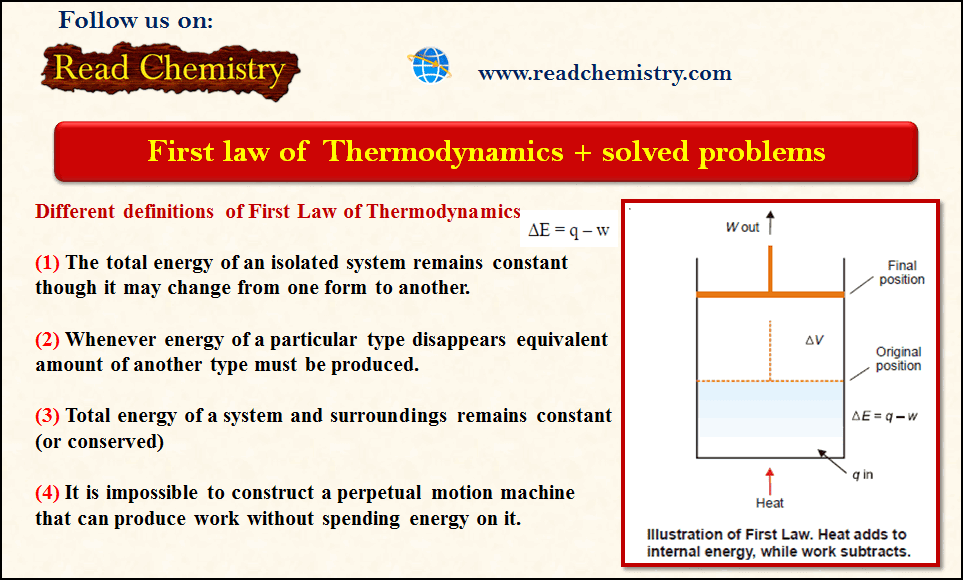
The First Law of Thermodynamics + Solved Problems
– The first law of Thermodynamics states that The total energy of an isolated system remains constant though it may…
Read More » -
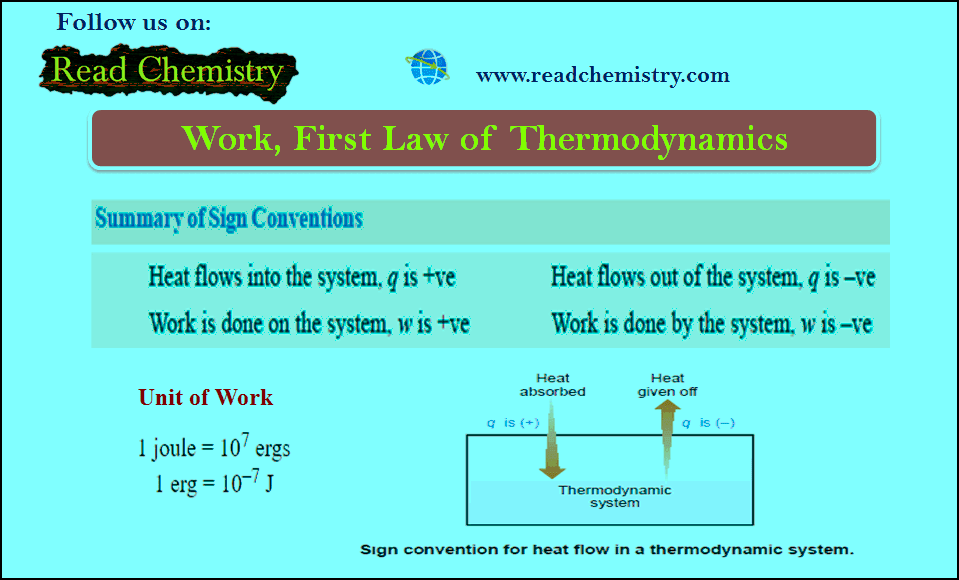
Work in Thermodynamics (Definition – Formula – Problems)
– In physics, mechanical work is defined as force multiplied by the distance through which the force acts. – In…
Read More » -
Thermodynamic Processes
– Thermodynamic Processes involve the change of conditions (temperature, pressure, and volume). Thermodynamic Processes – When a thermodynamic system changes…
Read More »