Physical Chemistry blends chemistry with physics, studying matter’s behavior using concepts like thermodynamics, kinetics, quantum mechanics, and spectroscopy. It explains how and why chemical reactions occur at the molecular level.
Physical Chemistry
-
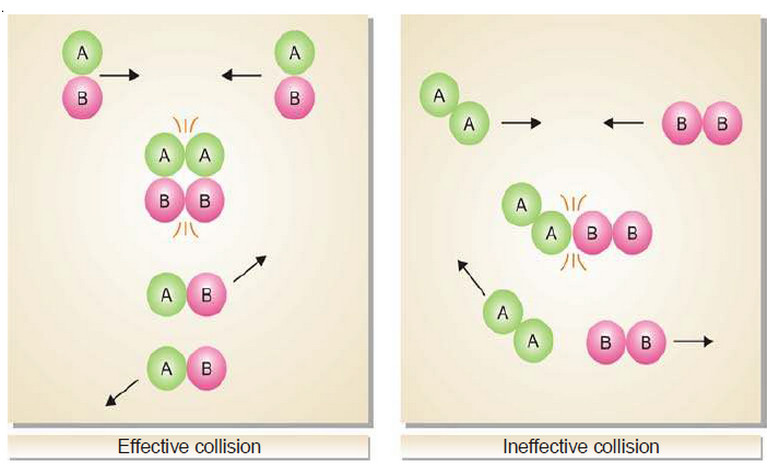
Collision theory of Reaction rates
Collision theory of Reaction rates – According to collision theory, a chemical reaction takes place only by collisions between the…
Read More » -
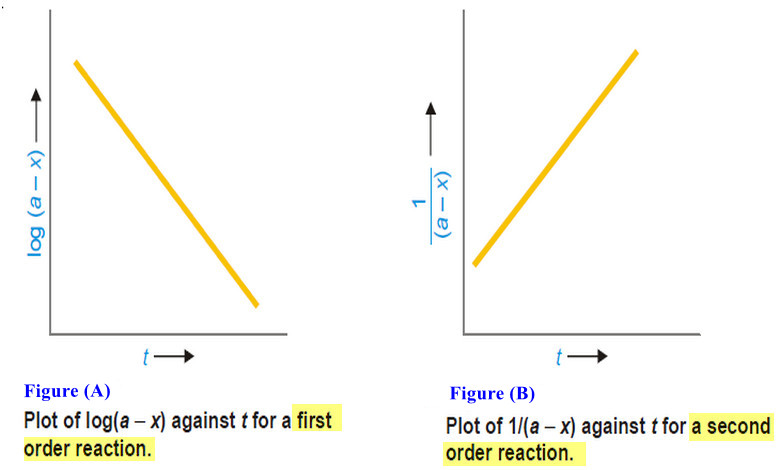
How to determine the order of reaction?
Determination of the order of reaction – There are at least four different methods to determine the order of reaction…
Read More » -
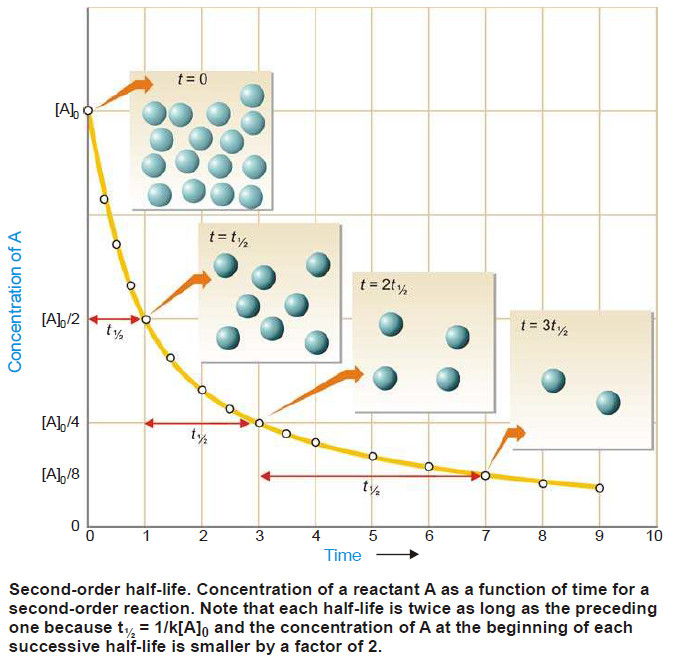
Second order reaction
Second order reaction – Let us take a second order reaction of the type 2A ⎯⎯→ products – Suppose the…
Read More » -
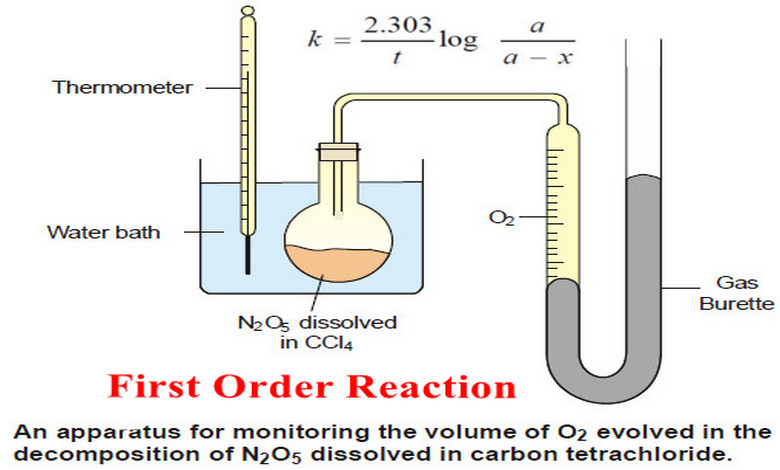
First Order Reaction -Examples and Solved problems
First order reaction – Let us consider a first order reaction: A → products – Suppose that at the beginning…
Read More » -
Molecular Vibrations : IR spectrum
– In this subject we talk about Molecular Vibrations as introduction to understand IR spectrum Molecular Vibrations – Before discussing…
Read More » -
Molecularity of a reaction
– we will discuss the Molecularity of a reaction and the Differences Between Order and Molecularity. Molecularity of a reaction…
Read More » -
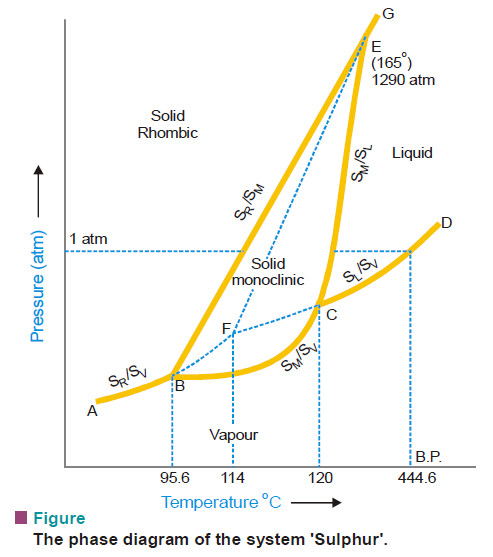
Sulphur system – Phase diagram of Sulphur
The Sulphur system – Sulphur system is a one-component, four-phase system. – The four phases are: (a) Two solid polymorphic…
Read More » -
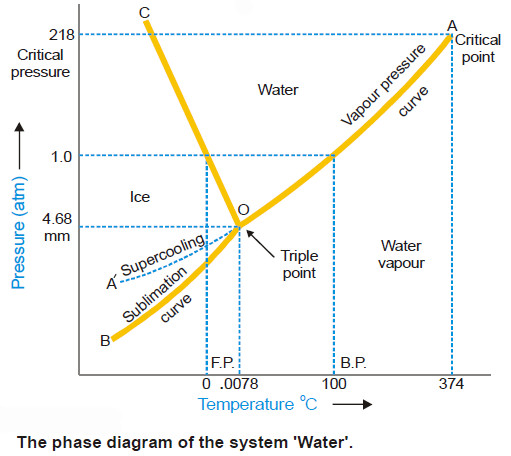
Water system, Phase diagram of Water
The Water system – Under normal conditions the Water system is a three-phase, one-component system. – The three phases involved…
Read More » -
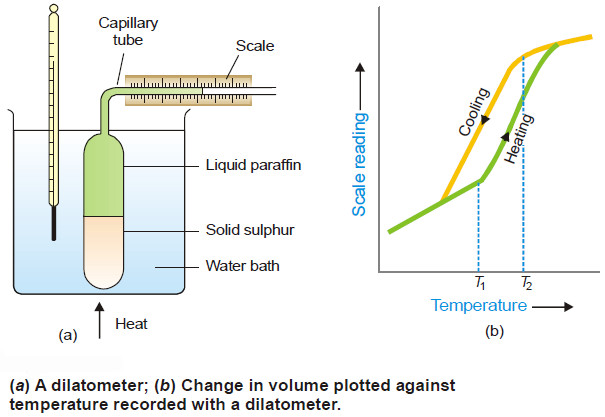
Polymorphism – Allotropy
Polymorphism – The occurrence of the same substance in more than one crystalline forms is known as Polymorphism. – Polymorphism…
Read More » -
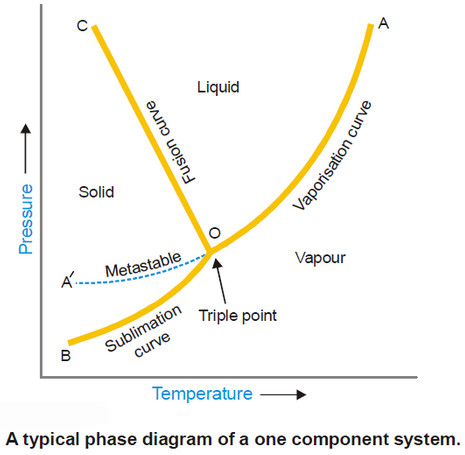
Phase diagram – Phase Rule
Phase Diagram – A phase diagram is a plot showing the conditions of pressure and temperature under which two or…
Read More » -
Derivation of the phase rule
Derivation of the phase rule – Here the derivation of the phase rule for one-component system and two-component system are…
Read More » -
Degree of Freedom – phase Rule
Degree of Freedom – The term Degree of Freedom represented by F in the phase Rule equation (F = C…
Read More » -
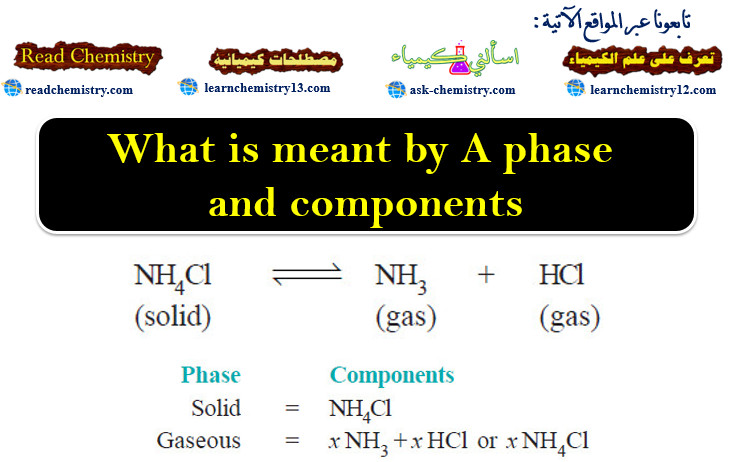
What is meant by A phase and components?
The Phase Rule statement – The phase Rule is an important generalization dealing with the behaviour of heterogeneous systems. –…
Read More » -
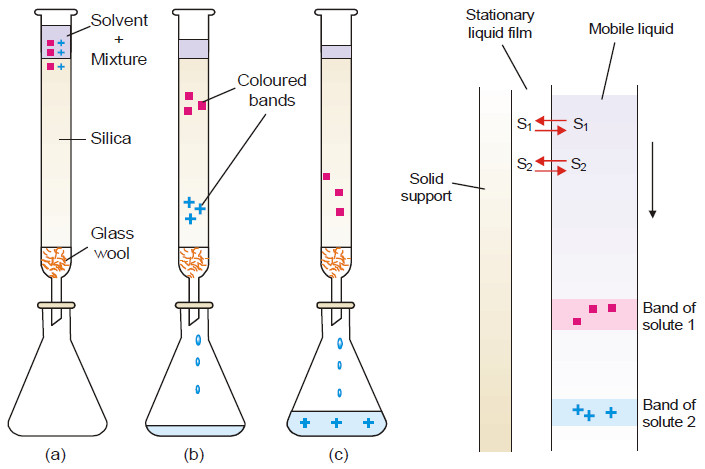
Applications of distribution law
Applications of distribution law – There are numerous applications of distribution law in the laboratory as well as in industry.…
Read More » -
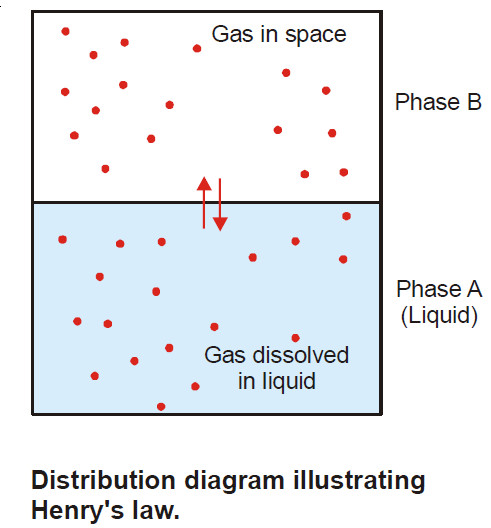
Henry’s law – a form of distribution law
Henry’s law statement – Henry’s law states: at a constant temperature the solubility of a gas in a liquid is…
Read More »