Physical Chemistry
Types of Radiations and its properties
Nuclear reaction
** A nuclear reaction is different from a chemical reaction.
** In a chemical reaction, atoms of the reactants combine by a rearrangement of extranuclear electrons but the nuclei of the atoms remain unchanged.
** In a nuclear reaction, on the other hand, it is the nucleus of the atom which is involved. The number of protons or neutrons in the nucleus changes to form a new element.
** A study of the nuclear changes in atoms is termed Nuclear Chemistry.
Radioactivity
** A number of elements such as uranium and radium are unstable. Their atomic nucleus breaks of its own accord to form a smaller atomic nucleus of another element. The protons and neutrons in the unstable nucleus regroup to give the new nucleus. This causes the release of excess particles and energy from the original nucleus, which we call radiation.
**The elements whose atomic nucleus emits radiation are said to be radioactive.
** The spontaneous breaking down of the unstable atoms is termed radioactive disintegration or radioactive decay.
**The disintegration or decay of unstable atoms accompanied by emission of radiation is called Radioactivity
Types of Radiations
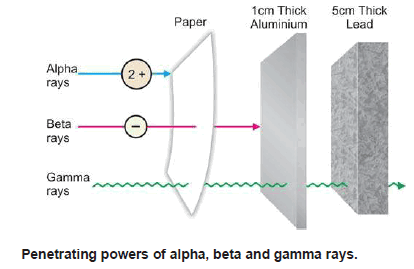
**The radioactive radiations are of three types. These were sorted out by Rutherford (1902) by passing them between two oppositely charged plates (Fig).
**The one bending towards the negative plate carried positive charge and were named α (alpha) rays. Those bending towards the positive plate and carrying negative charge were called β (beta) rays. The third type of radiation, being uncharged, passed straight through the electric field and were named γ (gamma) rays.
**α, β and γ rays could be easily detected as they cause luminescence on the zinc sulphide screen placed in their path.
Properties of Radiation
Alpha (α), beta (β) and gamma (γ) rays differ from each other in nature and properties. There chief properties are : (a) Velocity; (b) Penetrating power; (c) Ionization.
Alpha rays (α)
(1) Nature: They consist of streams of α-particles. By measurement of their e/m, Rutherford showed that they have a mass of 4 amu and charge of +2. They are helium nuclei and may be represented as 4α2or 4He2 .
(2) Velocity: α-particles are ejected from radioactive nuclei with very high velocity, about one-tenth that of light.
(3) Penetrating power: Because of their charge and relatively large size, α-particles have very little power of penetration through matter. They are stopped by a sheet of paper, 0.01 mm thick aluminium foil or a few centimeters of air.
(4) Ionization: city and attraction for electrons, α-particles break away electrons from gas molecules and convert them to positive ions.
Beta rays (β)
(1) Nature: They are streams of β-particles emitted by the nucleus. From their deflection electric and magnetic fields, Becquerel showed that β-particles are identical with electrons. They have very small mass (1/1827 amu) and charge of – 1. A β-particle is symbolized as 0β–1 or 0e–1.
(2) Velocity: They travel about 10 times faster than α-particles. Their velocity is about the same as of light.
(3) Penetrating power: β-Particles are 100 times more penetrating in comparison to α-particles. This is so because they have higher velocity and negligible mass. β-particles can be stopped by about 1 cm thick sheet of aluminium or 1 m of air.
(4) Ionization: The ionization produced by β-particles in a gas is about one-hundredth of that of α-particles. Though the velocity of β-particles is higher but the mass being smaller, their kinetic energy is much less than α-particles. Hence they are poor ionizers.
Gamma rays (γ)
(1) Nature: Unlike α and β-rays, they do not consist of particles of matter. γ-Rays are a form of electromagnetic radiation of shorter wavelength than X-rays. They could be thought of as high-energy photons released by the nucleus during α– or β-emissions. They have no mass or charge and may be symbolized as 0γ0
(2) Velocity: Like all forms of electromagnetic radiation, γ-rays travel with the velocity of light.
(3) Ionizing power: Their ionizing power is very weak in comparison to α– and β-particles. A γ-photon displaces an electron of the gas molecule to yield a positive ion. Since the chances of photon-electron collisions are small, γ-rays are weak ionizers.
(4) Penetrating power: Because of their high velocity and non-material nature, γ-rays are most penetrating. They cannot be stopped even by a 5 cm thick sheet of lead or several meters thick layer of concrete.
Comparison of properties of α, β and γ-rays
Artificial Radioactivity
**Many stable nuclei when bombarded with high speed particles produce unstable nuclei that are radioactive.
**The radioactivity produced in this manner by artificial means is known as artificial radioactivity or induced radioactivity.
**The artificial isotopes disintegrate in a definite fashion and have specific half-life.
**For example, aluminium-27 when bombarded with a neutron emits an alpha particle and forms sodium-24 which is radioactive. It disintegrates spontaneously by emission of a beta particle (0e–1) and the product is magnesium-24. Sodium-24 has half-life of 24 hours.
Nuclear Isomerism
**Sometimes α and β-decays may produce a pair of nuclei that have the same number of protons and neutrons but different radioactive properties.
**A pair of nuclei having same number of protons and neutrons but different half-lives are called nuclear isomers. The phenomenon is called nuclear isomerism.
**The nuclear isomers may be isotopic or isobaric
Example of Nuclear isomerism
Uranium-Z and Uranium-X2 constitute a pair of nuclear isomers. Both nuclei contain 91 protons and 143 neutrons, and are isotopes. They exhibit β-ray activity with half-lives 6.7 hr and 1.14 min respectively.
Explanation
The α– or β-decay of a radionuclide first leaves it in an excited state. This is then converted into the ground state nucleus. The excited and the ground state nuclei thus produced are called nuclear isomers. The nuclear isomers may be isotopic or isobaric.
Reference: Essentials of Physical Chemistry /Arun Bahl, B.S Bahl and G.D. Tuli / multicolour edition.