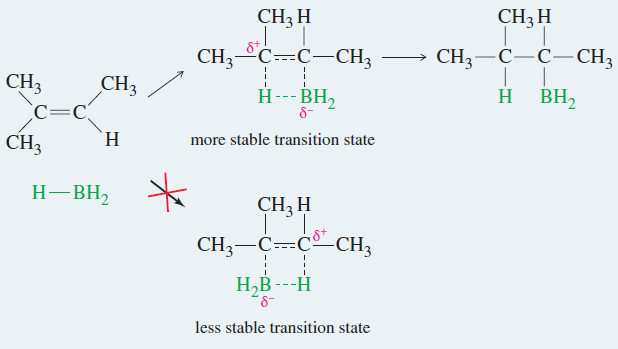
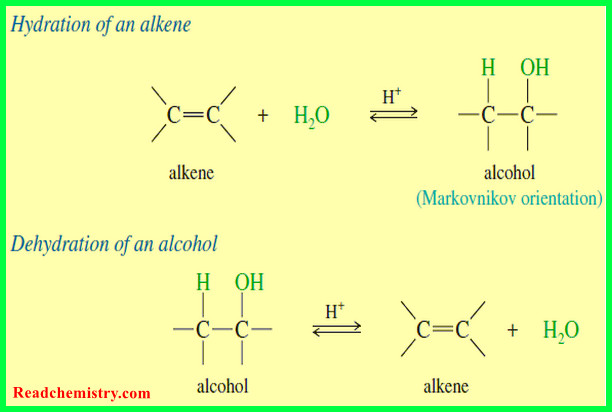
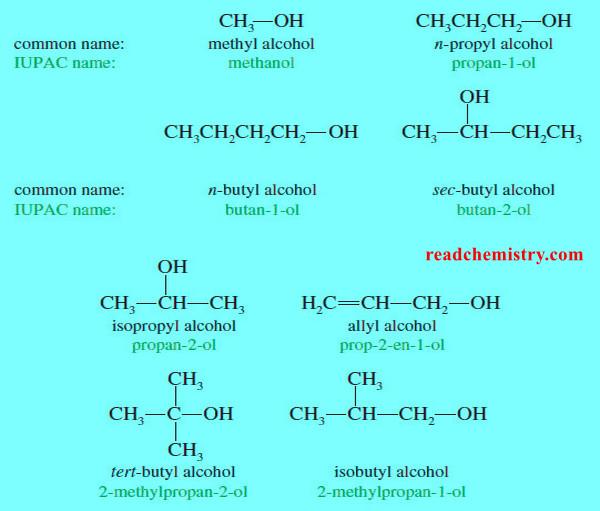
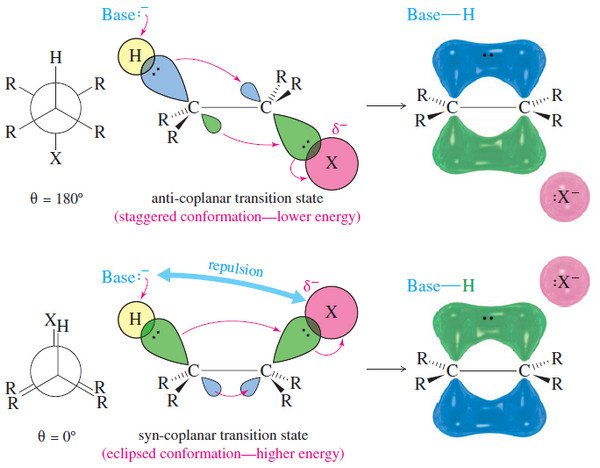
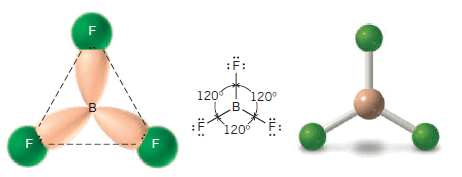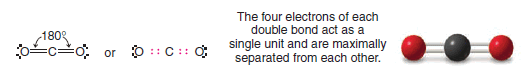Organic Chemistry
How To Predict Molecular Geometry: VSEPR model
How To Predict Molecular Geometry: VSEPR model
** We can predict the arrangement of atoms in molecules and ions on the basis of a relatively simple idea called the valence shell electron pair repulsion (VSEPR) model.
** We apply the VSEPR model in the following way:
(1) We consider molecules (or ions) in which the central atom is covalently bonded to two or more atoms or groups.
(2) We consider all of the valence electron pairs of the central atom—both those that are shared in covalent bonds, called bonding pairs, and those that are unshared, called nonbonding pairs or unshared pairs or lone pairs.
(3) Because electron pairs repel each other, the electron pairs of the valence shell tend to stay as far apart as possible. The repulsion between nonbonding pairs is generally greater than that between bonding pairs.
(4) We arrive at the geometry of the molecule by considering all of the electron pairs, bonding and nonbonding, but we describe the shape of the molecule or ion by referring to the positions of the nuclei (or atoms) and not by the positions of the electron pairs
In the following sections we consider several examples.
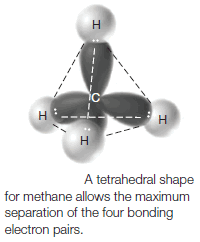
(1) Methane
** The valence shell of methane contains four pairs of bonding electrons.
** Only a tetrahedral orientation will allow four pairs of electrons to have equal and maximum possible separation from each other.
** Any other orientation, for example, a square planar arrangement, places some electron pairs closer together than others. Thus, methane has a tetrahedral shape.
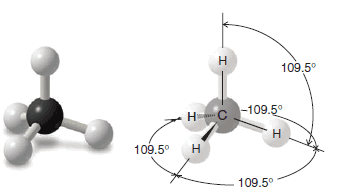
** The bond angles for any atom that has a regular tetrahedral structure are 109.5o. A representation of these angles in methane is shown in the following figure.
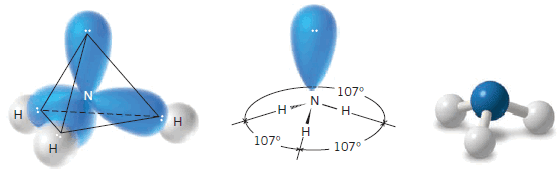
(2) Ammonia
** The shape of a molecule of ammonia (NH3) is a trigonal pyramid. There are three bonding pairs of electrons and one nonbonding pair.
** The bond angles in a molecule of ammonia are 107o, a value very close to the tetrahedral angle (109.5o).
** We can write a general tetrahedral structure for the electron pairs of ammonia by placing the nonbonding pair at one corner .
** A tetrahedral arrangement of the electron pairs explains the trigonal pyramidal arrangement of the four atoms.
** The bond angles are 107o (not 109.5o) because the nonbonding pair occupies more space than the bonding pairs.
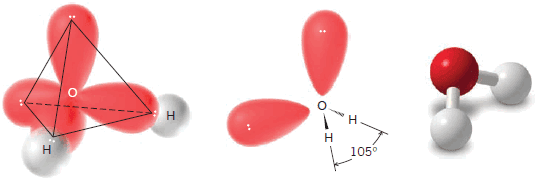
(3) Water
** A molecule of water has an angular or bent shape.
** The H-O-H bond angle in a molecule of water is 104.5o, an angle that is also quite close to the 109.5o bond angles of methane.
** We can write a general tetrahedral structure for the electron pairs of a molecule of water if we place the two bonding pairs of electrons and the two nonbonding electron pairs at the corners of the tetrahedron. Such a structure is shown in Fig. 1.36.
** A tetrahedral arrangement of the electron pairs accounts for the angular arrangement of the three atoms.
** The bond angle is less than 109.5o because the nonbonding pairs are effectively “larger” than the bonding pairs and, therefore, the structure is not perfectly tetrahedral.
(4) Boron Trifluoride
** Boron, a group IIIA element, has only three valence electrons. In the compound boron trifluoride (BF3) these three electrons are shared with three fluorine atoms.
** As a result, the boron atom in BF3 has only six electrons (three bonding pairs) around it. Maximum separation of three bonding pairs occurs when they occupy the corners of an equilateral triangle.
** Consequently, in the boron trifluoride molecule the three fluorine atoms lie in a plane at the corners of an equilateral triangle . Boron trifluoride is said to have a trigonal planar structure.
** The bond angles are 120o
(5) Beryllium Hydride
** The central beryllium atom of BeH2 has only two electron pairs around it; both electron pairs are bonding pairs.
** These two pairs are maximally separated when they are on opposite sides of the central atom, as shown in the following structures. This arrangement of the electron pairs accounts for the linear geometry of the BeH2 molecule .
** The bond angles are 180o.
(6) Carbon Dioxide
** The VSEPR method can also be used to predict the shapes of molecules containing multiple bonds if we assume that all of the electrons of a multiple bond act as though they were a single unit and, therefore, are located in the region of space between the two atoms joined by a multiple bond.
** This principle can be illustrated with the structure of a molecule of carbon dioxide (CO2).
** The central carbon atom of carbon dioxide is bonded to each oxygen atom by a double bond. Carbon dioxide is known to have a linear shape; the bond angle is 180o .
** Such a structure is consistent with a maximum separation of the two groups of four bonding electrons. The nonbonding pairs associated with the oxygen atoms have no effect on the shape.
Shapes of Molecules and Ions from VSEPR Theory
** The shapes of several simple molecules and ions as predicted by VSEPR theory are shown in the following Table:
** In this table we have also included the hybridization state of the central atom.
Reference: Organic chemistry / T.W. Graham Solomons , Craig B.Fryhle , Scott A.snyder , / ( eleventh edition) / 2014.