Physical Chemistry book , 3rd edition by Robert G. Mortimer
– In this subject, we will discuss free download of Physical Chemistry book, 3rd edition by Robert G. Mortimer

The Preface of Physical Chemistry book
– The book is divided into four parts:
– The first part focuses on the macroscopic properties of physical systems. It begins with the descriptive study of gases and liquids and proceeds to the study of thermodynamics, which is a comprehensive macroscopic theory of the behavior of material systems.
– The second part focuses on dynamics, including gas kinetic theory, transport processes, and chemical reaction kinetics.
– The third part presents quantum mechanics and spectroscopy.
– The fourth part presents the relationship between molecular and macroscopic properties of systems through the study of statistical mechanics. This theory is applied to the structure of condensed phases.
– The book is designed so that the first three parts can be studied in any order, while the fourth part is designed to be a capstone in which the other parts are integrated into a cohesive whole.
– In addition to the standard tables of integrals and numerical values of various properties, the book contains several appendices that expand on discussions in the body of the text, such as more detailed discussions of perturbation theory, group theory, and several mathematical topics.
– Each chapter begins with a statement of the principal facts and ideas that are presented in the chapter. There is a summary at the end of each chapter to assist in synthesizing the material of each chapter into a coherent whole.
– There are also marginal notes throughout the chapters that present biographical information and some comments.
– Each chapter contains examples that illustrate various kinds of calculations, as well as exercises placed within the chapter.
– Both these exercises and the problems at the end of each section are designed to provide practice in applying techniques and insights obtained through the study of the chapter.
– Answers to all of the numerical exercises and the odd-numbered numerical problems are placed in Appendix K.
Description of Physical Chemistry book
Book name: Physical Chemistry, 3rd edition by Robert G. Mortimer.
Author:
- Robert G. Mortimer
- Professor Emeritus
- Rhodes College
- Memphis, Tennessee
Edition: 3rd edition
Year: 2008
No of Chapters: 28
No of Pages: 1405
File format: pdf
File size: 10.5 MB
Contents of Physical Chemistry book
PART (I): Thermodynamics and the Macroscopic Description of Physical Systems
Chapter 1: The Behavior of Gases and Liquids
Chapter 2: Work, Heat, and Energy: The First Law of Thermodynamics
Chapter 3: The Second and Third Laws of Thermodynamics: Entropy
Chapter 4: The Thermodynamics of Real Systems
Chapter 5: Phase Equilibrium
Chapter 6: The Thermodynamics of Solutions
Chapter 7: Chemical Equilibrium
Chapter 8: The Thermodynamics of Electrochemical Systems
PART (II): Dynamics
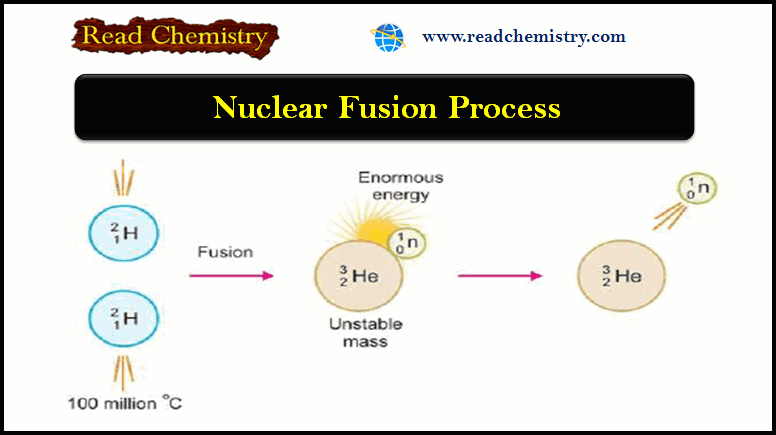
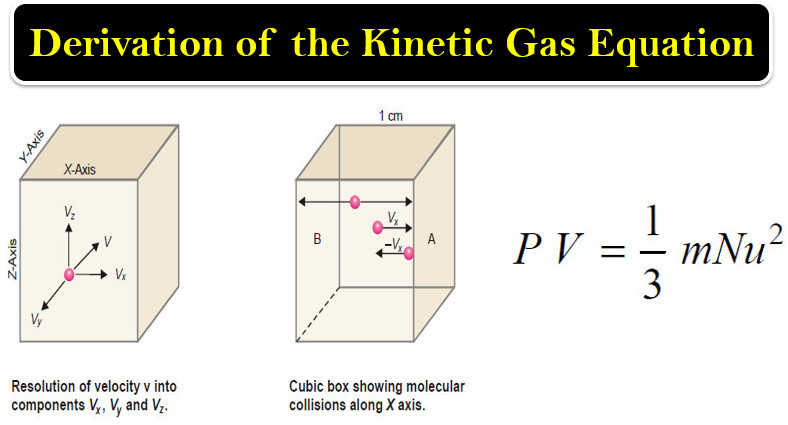
Chapter 9: Gas Kinetic Theory: The Molecular Theory of Dilute Gases at Equilibrium
Chapter 10: Transport Processes
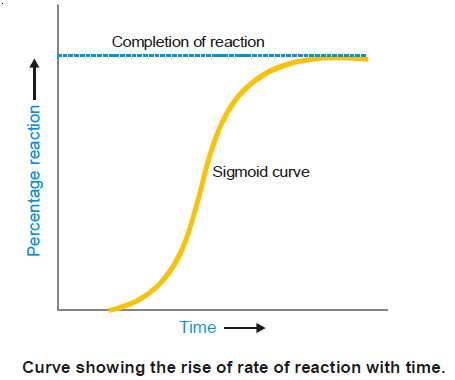
Chapter 11: The Rates of Chemical Reactions
Chapter 12: Chemical Reaction Mechanisms I: Rate Laws and Mechanisms
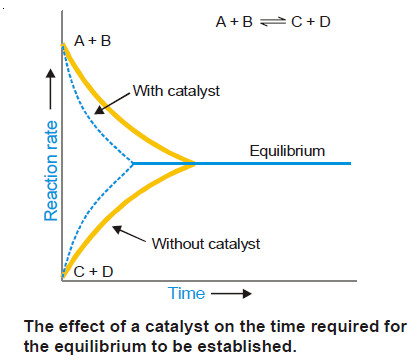
Chapter 13: Chemical Reaction Mechanisms II: Catalysis and Miscellaneous Topics
PART (III): The Molecular Nature of Matter
Chapter 14: Classical Mechanics and the Old Quantum Theory
Chapter 15: The Principles of Quantum Mechanics. I. De BroglieWaves and the Schrodinger Equation
Chapter 16: The Principles of Quantum Mechanics. II. The Postulates of Quantum Mechanics
Chapter 17: The Electronic States of Atoms. I. The Hydrogen Atom
Chapter 18: The Electronic States of Atoms. II.The Zero-OrderApproximation for Multielectron Atoms
Chapter 19: The Electronic States of Atoms. III. Higher-Order Approximations
Chapter 20: The Electronic States of Diatomic Molecules
Chapter 21: The Electronic Structure of Polyatomic Molecules
Chapter 22: Translational, Rotational, and Vibrational States of Atoms and Molecules
Chapter 23: Optical Spectroscopy and Photochemistry
Chapter 24: Magnetic Resonance Spectroscopy
PART (IV): The Reconciliation of the Macroscopic and Molecular Theories of Matter
Chapter 25: Equilibrium Statistical Mechanics I. The Probability Distribution for Molecular States
Chapter 26: Equilibrium Statistical Mechanics. II. Statistical Thermodynamics
Chapter 27: Equilibrium Statistical Mechanics. III. Ensembles
Chapter 28: The Structure of Solids, Liquids, and Polymers
Cover of the book
Free Download of Physical Chemistry book
File format: pdf
File size: 10.5 MB
- For more free books. Visit Category: Free books