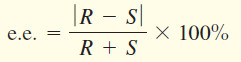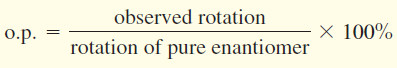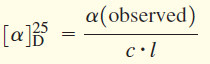Essential terms in Stereochemistry
Essential terms in Stereochemistry
stereochemistry
– stereochemistry is The study of the three-dimensional structure of molecules.
– It is the systematic presentation of a specific field of science and technology that traditionally requires a short preliminary excursion into history.
– Stereochemistry is the (chemistry of space).
– In this subject we will discuss essential terms in Stereochemistry
stereoisomers
– (configurational isomers) Isomers whose atoms are bonded together in the same order but differ in how the atoms are oriented in space.
– structural isomers (see constitutional isomers) Isomers that differ in the order in which their atoms are bonded together.
superimposable
– Identical in all respects.
– The three-dimensional positions of all atoms coincide when the molecules are placed on top of each other.
trans
On opposite sides of a ring or double bond.
absolute configuration
– The detailed stereochemical picture of a molecule, including how the atoms are arranged in space.
– Alternatively, the (R) or (S) configuration at each asymmetric carbon atom.
allenes
– Compounds having two C=C double bonds that meet at a single carbon atom, C=C=C
– The two outer carbon atoms are trigonal planar, with their planes perpendicular to each other.
– Many substituted allenes are chiral.
asymmetric carbon atom
(chiral carbon atom) A carbon atom that is bonded to four different groups.
Cahn–Ingold–Prelog convention
The accepted method for designating the absolute configuration of a chirality center (usually an asymmetric carbon) as either (R) or (S).
chiral
Different from its mirror image.
chiral carbon atom
(asymmetric carbon atom) A carbon atom that is bonded to four different groups.
chirality center
– (chiral center) The IUPAC term for an atom holding a set of ligands in a spatial arrangement that is not superimposable on its mirror image.
– Asymmetric carbon atoms are the most common chirality centers.
chiral probe
A molecule or an object that is chiral and can use its own chirality to differentiate between mirror images.
cis
On the same side of a ring or double bond.
cis-trans isomers
(geometric isomers) Isomers that differ in their geometric arrangement on a ring or double bond; cis-trans isomers are a subclass of diastereomers.
configurations
The two possible spatial arrangements around a chirality center or other stereocenter.
configurational isomers
(see stereoisomers)
conformers
– (conformational isomers) Structures that differ only by rotations about single bonds.
– In most cases, conformers interconvert at room temperature; thus, they are not different compounds and not true isomers.
constitutional isomers
(structural isomers) Isomers that differ in the order in which their atoms are bonded together.
D–L configurations
(Fischer–Rosanoff convention) D has the same relative configuration as (+)-glyceraldehyde.
L has the same relative configuration as (-)-glyceraldehyde.
dextrorotatory, (+) or (d)
Rotating the plane of polarized light clockwise.
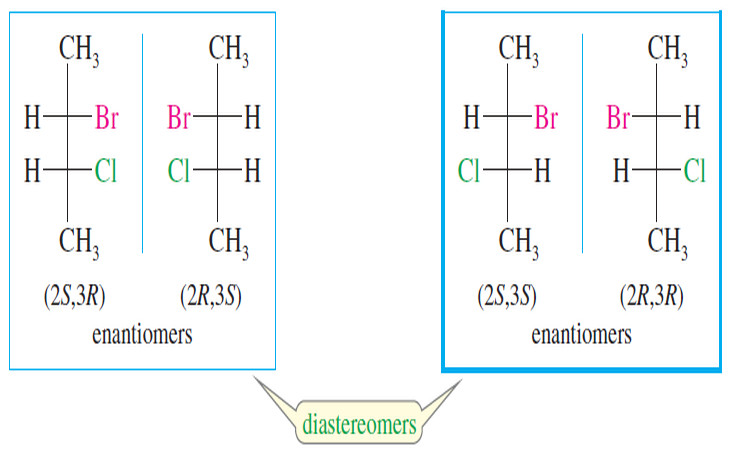
diastereomers
Stereoisomers that are not mirror images.
enantiomeric excess (e.e.)
– The excess of one enantiomer in a mixture of enantiomers expressed as a percentage of the mixture.
– Similar to optical purity. Algebraically,
enantiomers
A pair of nonsuperimposable mirror-image molecules: mirror-image isomers. (p. 176)
Fischer projection
– A method for drawing an asymmetric carbon atom as a cross.
– The carbon chain is kept along the vertical, with the IUPAC numbering from top to bottom.
– Vertical bonds project away from the viewer, and horizontal bonds project toward the viewer.
geometric isomers
(see cis-trans isomers)
internal mirror plane (σ)
– A plane of symmetry through the middle of a molecule, dividing the molecule into two mirror-image halves.
– A molecule with an internal mirror plane of symmetry cannot be chiral.
isomers
Different compounds with the same molecular formula.
Leftorium
An imaginary store that sells the enantiomers of everyday chiral objects such as scissors, rifles, can openers, etc.
levorotatory, (-) or (l)
Rotating the plane of polarized light counterclockwise.
meso compound
– An achiral compound that contains chirality centers (usually asymmetric carbon atoms).
– Originally, an achiral compound that has chiral diastereomers.
optical isomers
(archaic; see enantiomers) Compounds with identical properties except for the direction in which they rotate polarized light.
optical activity
Rotation of the plane of polarized light.
optically active
Capable of rotating the plane of polarized light.
optical purity (o.p.)
– The specific rotation of a mixture of two enantiomers, expressed as a percentage of the specific rotation of one of the pure enantiomers.
– Similar to enantiomeric excess.
– Algebraically,
plane-polarized light
Light composed of waves that vibrate in only one plane.
polarimeter
An instrument that measures the rotation of plane-polarized light by an optically active compound.
racemic mixture
[racemate, racemic modification, (±) pair, (d,l) pair] A mixture of equal quantities of enantiomers, such that the mixture is optically inactive.
relative configuration
The experimentally determined relationship between the configurations of two molecules, even though the absolute configuration of either may not be known.
resolution
The process of separating a racemic mixture into the pure enantiomers. Resolution requires a chiral resolving agent.
resolving agent
A chiral compound (or chiral material on a chromatographic column) used for separating enantiomers.
2n rule
A molecule with (n) chiral carbon atoms might have as many as stereoisomers.
specific rotation
A measure of a compound’s ability to rotate the plane of polarized light, given by:
where c is concentration in g/mL and l is length of sample cell (path length) in decimeters.
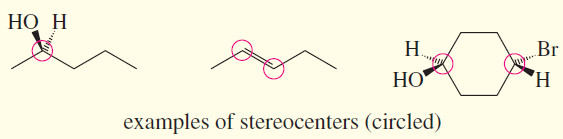
stereocenter
– (stereogenic atom) An atom that gives rise to stereoisomers when its groups are interchanged.
– Asymmetric carbon atoms and double-bonded carbons in cis-trans alkenes are the most common stereocenters.