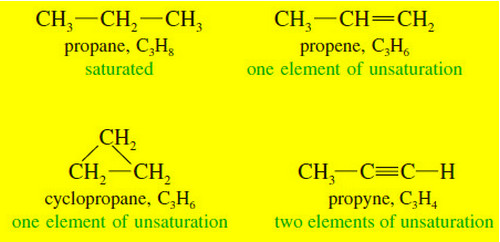
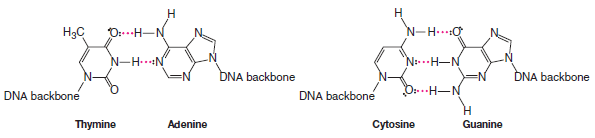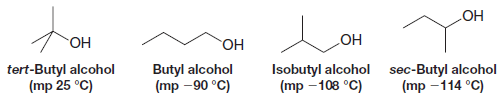Organic Chemistry
Physical Properties and Molecular Structure of Organic compound
** So far, we have said little about one of the most obvious characteristics of organic compounds— that is, their physical state or phase. Whether a particular substance is a solid, or a liquid, or a gas would certainly be one of the first observations that we would note in any experimental work. The temperatures at which transitions occur between phases— that is, melting points (mp) and boiling points (bp)—are also among the more easily measured physical properties. Melting points and boiling points are also useful in identifying and isolating organic compounds.
** Suppose, for example, we have just carried out the synthesis of an organic compound that is known to be a liquid at room temperature and 1 atm pressure. If we know the boiling point of our desired product and the boiling points of by-products and solvents that may be present in the reaction mixture, we can decide whether or not simple distillation will be a feasible method for isolating our product. In another instance our product might be a solid. In this case, in order to isolate the substance by crystallization, we need to know its melting point and its solubility in different solvents.
** The physical constants of known organic substances are easily found in handbooks and other reference books. Table (1) lists the melting and boiling points of some of the compounds.
 |
| Table (1) |
** Often in the course of research, however, the product of a synthesis is a new compound— one that has never been described before. In these instances, success in isolating the new compound depends on making reasonably accurate estimates of its melting point, boiling point, and solubilities. Estimations of these macroscopic physical properties are based on the most likely structure of the substance and on the forces that act between molecules and ions. The temperatures at which phase changes occur are an indication of the strength of these intermolecular forces.
(1) Ionic Compounds: Ion–Ion Forces
** The melting point of a substance: is the temperature at which an equilibrium exists between the well-ordered crystalline state and the more random liquid state.
** If the substance is an ionic compound, such as sodium acetate (Table 1), the ion–ion forces that hold the ions together in the crystalline state are the strong electrostatic lattice forces that act between the positive and negative ions in the orderly crystalline structure.
** In Fig (1) each sodium ion is surrounded by negatively charged acetate ions, and each acetate ion is surrounded by positive sodium ions.
** A large amount of thermal energy is required to break up the orderly structure of the crystal into the disorderly open structure of a liquid. As a result, the temperature at which sodium acetate melts is quite high, 324 oC.
** The boiling points of ionic compounds are higher still, so high that most ionic organic compounds decompose (are changed by undesirable chemical reactions) before they boil. Sodium acetate shows this behavior.
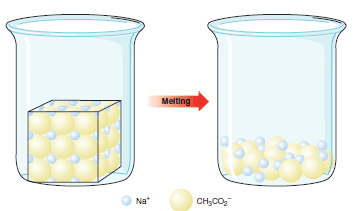 |
| Fig (1): The melting of Sodium acetate |
(2) Intermolecular Forces (van der Waals Forces)
** The forces that act between molecules are not as strong as those between ions, but they account for the fact that even completely nonpolar molecules can exist in liquid and solid states.These intermolecular forces, collectively called van der Waals forces, are all electrical in nature.
** We will focus our attention on three types:
(1) Dipole–dipole forces
(2) Hydrogen bonds
(3) Dispersion forces
(A) Dipole–Dipole Forces
** Most organic molecules are not fully ionic but have instead a permanent dipole moment resulting from a nonuniform distribution of the bonding electrons.
** Acetone and acetaldehyde are examples of molecules with permanent dipoles because the carbonyl group that they contain is highly polarized. In these compounds, the attractive forces between molecules are much easier to visualize.
** In the liquid or solid state, dipole–dipole attractions cause the molecules to orient themselves so that the positive end of one molecule is directed toward the negative end of another (Fig. 2).
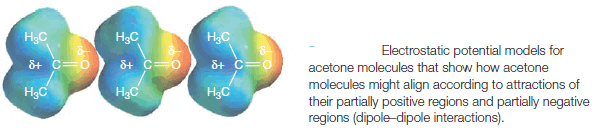 |
| Figure (2) |
(B) Hydrogen Bonds
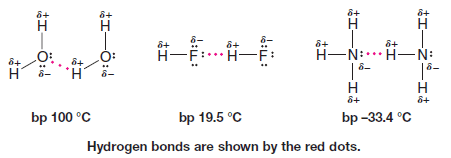
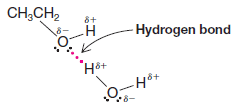
** Very strong dipole–dipole attractions occur between hydrogen atoms bonded to small, strongly electronegative atoms (O, N, or F) and nonbonding electron pairs on other such electronegative atoms. This type of intermolecular force is called a hydrogen bond.
** Hydrogen bonds (bond dissociation energies of about 4 – 38 kJ mol-1) are weaker than ordinary covalent bonds but much stronger than the dipole–dipole interactions that occur above, for example, in acetone.
** Hydrogen bonding explains why water, ammonia, and hydrogen fluoride all have far higher boiling points than methane (bp -161.6 oC), even though all four compounds have similar molecular weights.
** One of the most important consequences of hydrogen bonding is that it causes water to be a liquid rather than a gas at 25 oC. Calculations indicate that in the absence of hydrogen bonding, water would have a boiling point near -80 oC and would not exist as a liquid unless the temperature were lower than that temperature. Had this been the case, it is highly unlikely that life, as we know it, could have developed on the planet Earth.
** Hydrogen bonds hold the base pairs of double-stranded DNA together. Thymine hydrogen bonds with adenine. Cytosine hydrogen bonds with guanine.
** Hydrogen bonding accounts for the fact that ethyl alcohol has a much higher boiling point (78.5 oC) than dimethyl ether (24.9 oC) even though the two compounds have the same molecular weight. Molecules of ethyl alcohol, because they have a hydrogen atom covalently bonded to an oxygen atom, can form strong hydrogen bonds to each other.
** Molecules of dimethyl ether, because they lack a hydrogen atom attached to a strongly electronegative atom, cannot form strong hydrogen bonds to each other. In dimethyl ether the intermolecular forces are weaker dipole–dipole interactions
** A factor (in addition to polarity and hydrogen bonding) that affects the melting point of many organic compounds is the compactness and rigidity of their individual molecules.
** Molecules that are symmetrical generally have abnormally high melting points. tert-Butyl alcohol, for example, has a much higher melting point than the other isomeric alcohols shown here:
(C) Dispersion Forces
** If we consider a substance like methane where the particles are nonpolar molecules, we find that the melting point and boiling point are very low: -182.6 oC and -162 oC, respectively.
** Instead of asking, “Why does methane melt and boil at low temperatures?” a more appropriate question might be “Why does methane, a nonionic, nonpolar substance, become a liquid or a solid at all?” The answer to this question can be given in terms of attractive intermolecular forces called dispersion forces or London forces.
** An accurate account of the nature of dispersion forces requires the use of quantum mechanics.
** We can, however, visualize the origin of these forces in the following way:
(1) The average distribution of charge in a nonpolar molecule (such as methane) over a period of time is uniform.
(2) At any given instant, however, because electrons move, the electrons and therefore the charge may not be uniformly distributed.
(3) Electrons may, in one instant, be slightly accumulated on one part of the molecule, and, as a consequence, a small temporary dipole will occur (Fig. 3).
 |
| Figure (3) |
(4) This temporary dipole in one molecule can induce opposite (attractive) dipoles in surrounding molecules. It does this because the negative (or positive) charge in a portion of one molecule will distort the electron cloud of an adjacent portion of another molecule, causing an opposite charge to develop there.
(5) These temporary dipoles change constantly, but the net result of their existence is to produce attractive forces between nonpolar molecules and thus make possible the existence of their liquid and solid states.
** Two important factors determine the magnitude of dispersion forces.
(1) The relative polarizability of electrons of the atoms involved.
** By polarizability we mean how easily the electrons respond to a changing electric field. The electrons of large atoms such as iodine are loosely held and are easily polarized, while the electrons of small atoms such as fluorine are more tightly held and are much less polarizable.
** CF4and CI4 are both nonpolar molecules. But if we were to consider the intermolecular forces between two CI4 molecules, which contain polarizable iodine atoms, we would find that the dispersion forces are much larger than between two CF4 molecules, which contains fluorine atoms that are not very polarizable.
(2) The relative surface area of the molecules involved.
** The larger the surface area, the larger is the overall attraction between molecules caused by dispersion forces. Molecules that are generally longer, flatter, or cylindrical have a greater surface area available for intermolecular interactions than more spherical molecules, and consequently have greater attractive forces between them than the tangential interactions between branched molecules.
** This is evident when comparing pentane, the unbranched C5H12hydrocarbon, with neopentane, the most highly branched C5H12isomer (in which one carbon bears four methyl groups). Pentane has a boiling point of 36.1 oC. Neopentane has a boiling point of 9.5 oC. The difference in their boiling points indicates that the attractive forces between pentane molecules are stronger than between neopentane molecules.
** For large molecules, the cumulative effect of these small and rapidly changing dispersion
forces can lead to a large net attraction.
(3) Boiling Points
** The boiling point of a liquid is the temperature at which the vapor pressure of the liquid equals the pressure of the atmosphere above it.
** The boiling points of liquids are pressure dependent, and boiling points are always reported as occurring at a particular pressure, at 1 atm (or at 760 torr), for example. A substance that boils at 150 oC at 1 atm pressure will boil at a substantially lower temperature if the pressure is reduced to, for example, 0.01 torr (a pressure easily obtained with a vacuum pump). The normal boiling point given for a liquid is its boiling point at 1 atm.
** In passing from a liquid to a gaseous state, the individual molecules (or ions) of the substance must separate. Because of this, we can understand why ionic organic compounds often decompose before they boil. The thermal energy required to completely separate (volatilize) the ions is so great that chemical reactions (decompositions) occur first.
** Nonpolar compounds, where the intermolecular forces are very weak, usually boil at low temperatures even at 1 atm pressure. This is not always true, however, because of other factors that we have not yet mentioned the effects of:
(1) molecular weight
(2) molecular shape and
(3) surface area.
** Heavier molecules require greater thermal energy in order to acquire velocities sufficiently great to escape the liquid phase, and because the surface areas of larger molecules can be much greater, intermolecular dispersion attractions can also be much larger.
** These factors explain why nonpolar ethane (bp -88.2 oC) boils higher than methane (bp -162 oC) at a pressure of 1 atm. It also explains why, at 1 atm, the even heavier and larger nonpolar molecule decane (C10H22) boils at 174 oC.
** The relationship between dispersion forces and surface area helps us understand why neopentane (2,2-dimethylpropane) has a lower boiling point (9.5 oC) than pentane (36.1 oC), even though they have the same molecular weight. The branched structure of neopentane allows less surface interaction between neopentane molecules, hence lower dispersion forces, than does the linear structure of pentane.
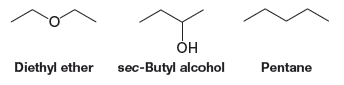
Solved Problem:Arrange the following compounds according to their expected boiling points, with the lowest boiling point first, and explain your answer. Notice that the compounds have similar molecular weights.
Strategy and Answer:
** Pentane has no polar groups and has only dispersion forces holding its molecules together. It would have the lowest boiling point.
** Diethyl ether has the polar ether group that provides dipole–dipole forces which are greater than dispersion forces, meaning it would have a higher boiling point than pentane.
** sec-Butyl alcohol has an -OH group that can form strong hydrogen bonds; therefore, it would have the highest boiling point.
(4) Solubility
** Intermolecular forces are of primary importance in explaining the solubilities of substances.
** Dissolution of a solid in a liquid is, in many respects, like the melting of a solid. The orderly crystal structure of the solid is destroyed, and the result is the formation of the more disorderly arrangement of the molecules (or ions) in solution.
** In the process of dissolving, too, the molecules or ions must be separated from each other, and energy must be supplied for both changes. The energy required to overcome lattice energies and intermolecular or interionic attractions comes from the formation of new attractive forces between solute and solvent.
** Consider the dissolution of an ionic substance as an example:
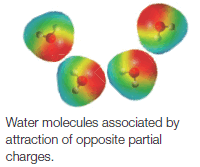
(1) Here both the lattice energy and interionic attractions are large. We find that water and only a few other very polar solvents are capable of dissolving ionic compounds. These solvents dissolve ionic compounds by hydrating or solvating the ions (Fig. 4).
 |
| Figure (4) |
(2) Water molecules, by virtue of their great polarity as well as their very small, compact shape, can very effectively surround the individual ions as they are freed from the crystal surface.
(3) Positive ions are surrounded by water molecules with the negative end of the water dipole pointed toward the positive ion; negative ions are solvated in exactly the opposite way.
(4) Because water is highly polar, and because water is capable of forming strong hydrogen bonds, the ion dipole forces of attraction are also large.
(5) The energy supplied by the formation of these forces is great enough to overcome both the lattice energy and interionic attractions of the crystal.
General rule for solubility
** A general rule for solubility is that “like dissolves like” in terms of comparable polarities:
(1) Polar and ionic solids are usually soluble in polar solvents.
(2) Polar liquids are usually miscible.
(3) Nonpolar solids are usually soluble in nonpolar solvents.
(4) Nonpolar liquids are usually miscible.
(5) Polar and nonpolar liquids, like oil and water, are usually not soluble to large extents
** Methanol and water are miscible in all proportions; so too are mixtures of ethanol and water and mixtures of both propyl alcohols and water. In these cases the alkyl groups of the alcohols are relatively small, and the molecules therefore resemble water more than they do an alkane.
** Another factor in understanding their solubility is that the molecules are capable of forming strong hydrogen bonds to each other:
Hydrophobic and Hydrophilic groups
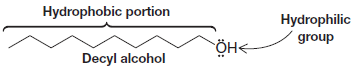
** We often describe molecules or parts of molecules as being hydrophilic or hydrophobic. The alkyl groups of methanol, ethanol, and propanol are hydrophobic. Their hydroxyl groups are hydrophilic.
(a) Hydrophobic:means incompatible with water (hydro, water; phobic, fearing or avoiding).
(b) Hydrophilic: means compatible with water (philic, loving or seeking).
** Decyl alcohol, with a chain of 10 carbon atoms, is a compound whose hydrophobic alkyl
group overshadows its hydrophilic hydroxyl group in terms of water solubility
** An explanation for why nonpolar groups such as long alkane chains avoid an aqueous
environment—that is, for the so-called hydrophobic effect—is complex.
** The most important factor seems to involve an unfavorable entropy change in the water. Entropy changes have to do with changes from a relatively ordered state to a more disordered one or the reverse.
** Changes from order to disorder are favorable, whereas changes from disorder to order are unfavorable. For a nonpolar hydrocarbon chain to be accommodated by water, the water molecules have to form a more ordered structure around the chain, and for this, the entropy change is unfavorable.
** The presence of a hydrophobic group and a hydrophilic group are essential components of soaps and detergents.
** The hydrophobic long carbon chains of a soap or detergent embed themselves in the oily layer that typically surrounds the thing we want to wash away.
** The hydrophilic ionic groups at the ends of the chains are then left exposed on the surface and make the surface one that water molecules find attractive.
** Oil and water don’t mix, but now the oily layer looks like something ionic and the water can take it “right down the drain.”
(5) Guidelines for Water Solubility
** Organic chemists usually define a compound as water soluble if at least 3 g of the organic compound dissolves in 100 mL of water.
** We find that for compounds containing one hydrophilic group—and thus capable of forming strong hydrogen bonds—the following approximate guidelines hold:
(1) compounds with one to three carbon atoms are water soluble,
(2) compounds with four or five carbon atoms are borderline
(3) compounds with six carbon atoms or more are insoluble.
Note
** When a compound contains more than one hydrophilic group, these guidelines do not apply.
** Polysaccharides, proteins and nucleic acids all contain thousands of carbon atoms and many are water soluble. They dissolve in water because they also contain thousands of hydrophilic groups.
(6) Intermolecular Forces in Biochemistry
** Later, after we have had a chance to examine in detail the properties of the molecules that make up living organisms, we shall see how intermolecular forces are extremely important in the functioning of cells.
** Hydrogen bond formation, the hydration of polar groups, and the tendency of nonpolar groups to avoid a polar environment all cause complex protein molecules to fold in precise ways—ways that allow them to function as biological catalysts of incredible efficiency.
** The same factors (above) allow molecules of hemoglobin to assume the shape needed to transport oxygen. They allow proteins and molecules called lipids to function as cell membranes.
** Hydrogen bonding gives certain carbohydrates a globular shape that makes them highly efficient food reserves in animals. It gives molecules of other carbohydrates a rigid linear shape that makes them perfectly suited to be structural components in plants.
(7) Summary of Attractive Electric Forces
The attractive forces occurring between molecules and ions that we have studied so far
are summarized in Table 2.
 |
| Table (2) |
Reference: Organic chemistry / T.W. Graham Solomons , Craig B.Fryhle , Scott A.snyder , / ( eleventh edition) / 2014.